 “Astrocytomas, the most prevalent primary brain tumors, can be divided by histology and malignancy levels into four following types: pilocytic astrocytoma (grade I), diffuse fibrillary astrocytoma (grade II), anaplastic astrocytoma (grade III), and glioblastoma multiforme (grade IV). For high grade astrocytomas (grade III and grade IV), blood vessels formation is considered as the most important property.
“Astrocytomas, the most prevalent primary brain tumors, can be divided by histology and malignancy levels into four following types: pilocytic astrocytoma (grade I), diffuse fibrillary astrocytoma (grade II), anaplastic astrocytoma (grade III), and glioblastoma multiforme (grade IV). For high grade astrocytomas (grade III and grade IV), blood vessels formation is considered as the most important property.
The distribution of cannabinoid receptors type 1 (CB1) and cannabinoid receptor type 2 (CB2) in blood vessels and tumor tissue of astrocytoma is still controversial. Asrocytoma tissues were collected from 45 patients under the condition of tumor-related neurosurgical operation. The expression of CB1 and CB2 receptors was assessed using immunofluorescence, quantitative real-time RT-PCR and western blotting.
The results indicated an increased expression of CB1 receptors in tumor tissue. There was a significant difference in the mount of CB2 receptors in blood vessels. More was observed in the grade III and glioblastoma (grade IV) than astrocytoma of grade II and control.
This study suggested that, the expression increase of cannabinoid receptors is an index for astrocytoma malignancy and can be targeted as a therapeutic approach for the inhibition of astrocytoma growth among patients.”
https://pubmed.ncbi.nlm.nih.gov/32623617/
https://link.springer.com/article/10.1007%2Fs11033-020-05636-8

 “Acute Respiratory Distress Syndrome (ARDS) is a life-threatening complication that can ensue following Staphylococcus aureus infection. The enterotoxin produced by these bacteria (SEB) acts as a superantigen thereby activating a large proportion of T cells leading to cytokine storm and severe lung injury.
“Acute Respiratory Distress Syndrome (ARDS) is a life-threatening complication that can ensue following Staphylococcus aureus infection. The enterotoxin produced by these bacteria (SEB) acts as a superantigen thereby activating a large proportion of T cells leading to cytokine storm and severe lung injury. “Multiple myeloma (MM) is characterized by aberrant bone marrow plasma cell (PC) proliferation and is one of the most common hematological malignancies. The potential effect of cannabinoids on the immune system and hematological malignancies has been poorly characterized.
“Multiple myeloma (MM) is characterized by aberrant bone marrow plasma cell (PC) proliferation and is one of the most common hematological malignancies. The potential effect of cannabinoids on the immune system and hematological malignancies has been poorly characterized.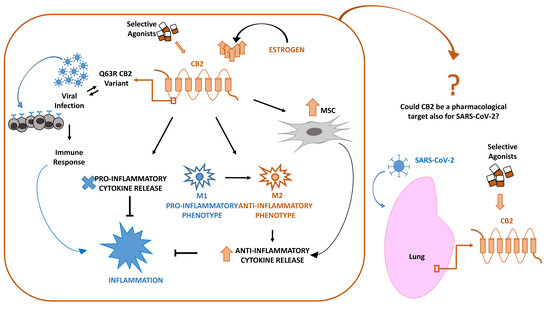
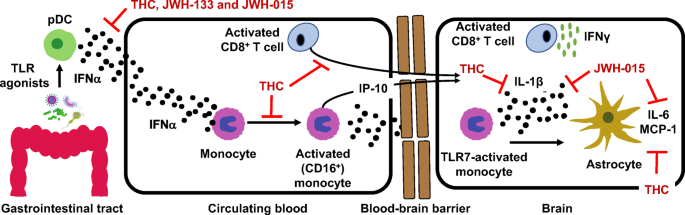
 “The antioxidant and CB2 receptor agonist properties of Δ9-tetrahydrocannabivarin (Δ9-THCV) afforded neuroprotection in experimental Parkinson’s disease (PD), whereas its CB1 receptor antagonist profile at doses lower than 5 mg/kg caused anti-hypokinetic effects.
“The antioxidant and CB2 receptor agonist properties of Δ9-tetrahydrocannabivarin (Δ9-THCV) afforded neuroprotection in experimental Parkinson’s disease (PD), whereas its CB1 receptor antagonist profile at doses lower than 5 mg/kg caused anti-hypokinetic effects. “Neuroinflammation in the rostral ventrolateral medulla (RVLM) has been reported to be associated with hypertension. The upregulation and activation of the
“Neuroinflammation in the rostral ventrolateral medulla (RVLM) has been reported to be associated with hypertension. The upregulation and activation of the  “Cannabis benefits patients with inflammatory bowel disease (IBD).
“Cannabis benefits patients with inflammatory bowel disease (IBD). “Microglia, the resident immune cells of the central nervous system, mediate brain homeostasis by controlling neuronal proliferation/differentiation and synaptic activity. In response to external signals from neuropathological conditions, homeostatic (M0) microglia can adopt one of two activation states: the classical (M1) activation state, which secretes mediators of the proinflammatory response, and the alternative (M2) activation state, which presumably mediates the resolution of neuroinflammation and tissue repair/remodeling.
“Microglia, the resident immune cells of the central nervous system, mediate brain homeostasis by controlling neuronal proliferation/differentiation and synaptic activity. In response to external signals from neuropathological conditions, homeostatic (M0) microglia can adopt one of two activation states: the classical (M1) activation state, which secretes mediators of the proinflammatory response, and the alternative (M2) activation state, which presumably mediates the resolution of neuroinflammation and tissue repair/remodeling. “Septic lung injury is one of main causes of high mortality in severe patients. Inhibition of excessive inflammatory response is considered as an effective strategy for septic lung injury.
“Septic lung injury is one of main causes of high mortality in severe patients. Inhibition of excessive inflammatory response is considered as an effective strategy for septic lung injury.