“Approximately one third of patients diagnosed with schizophrenia do not achieve adequate symptom control with standard antipsychotic drugs (APs).
The endocannabinoid system (ECS) in the brain plays an important role in maintaining normal mental health.
ECS modulates emotion, reward processing, sleep regulation, aversive memory extinction and HPA axis regulation…
The cannabis plant synthesises a large number of pharmacologically active compounds unique to it known as phytocannabinoids. In contrast to the euphoric and pro-psychotic effects of delta-9-tetrahydrocannabinol (THC), certain non-intoxicating phytocannabinoids have emerged in pre-clinical and clinical models as potential APs.
Since the likely mechanism of action does not rely upon dopamine D2 receptor antagonism, synergistic combinations with existing APs are plausible.
The anti-inflammatory and immunomodulatory effects of the non-intoxicating phytocannabinoid cannabidiol (CBD) are well established and are summarised below.
Preliminary data reviewed in this paper suggest that CBD in combination with a CB1 receptor neutral antagonist could not only augment the effects of standard APs but also target the metabolic, inflammatory and stress-related components of the schizophrenia phenotype.”
http://www.ncbi.nlm.nih.gov/pubmed/23829368
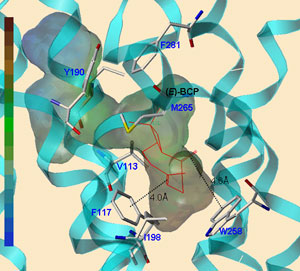
“6”-Azidohex-2″-yne-cannabidiol: a potential neutral, competitive cannabinoid CB1 receptor antagonist… 6″-azidohex-2″-yne-cannabidiol was as potent as cannabidiol in producing surmountable antagonism… it is a competitive cannabinoid CB(1) receptor antagonist…it may be a neutral cannabinoid CB(1) receptor antagonist.” http://www.ncbi.nlm.nih.gov/pubmed/15033394