“Major Depressive Disorder (MDD) is a highly prevalent and disabling psychiatric disorder, representing a major global health burden across all age groups.
Increasing evidence indicates that its pathophysiology involves a complex interplay between chronic stress, dysregulation of the hypothalamic-pituitary-adrenal (HPA) axis, immune activation, and neuroinflammation. Persistent HPA axis hyperactivity, glucocorticoid resistance, and altered expression of key regulators such as FKBP51 contribute to sustained inflammatory signaling and impaired neural plasticity in brain regions involved in mood regulation. Epigenetic mechanisms, including DNA methylation and microRNA-mediated regulation, further modulate stress responsivity, inflammatory pathways, and vulnerability to major depressive disorder.
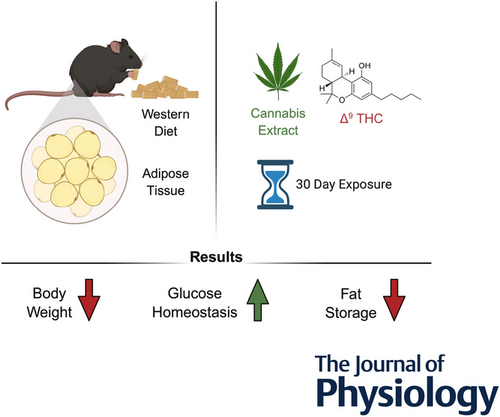
In this context, growing attention has been directed toward Cannabis sativa and its bioactive constituents as potential therapeutic agents.
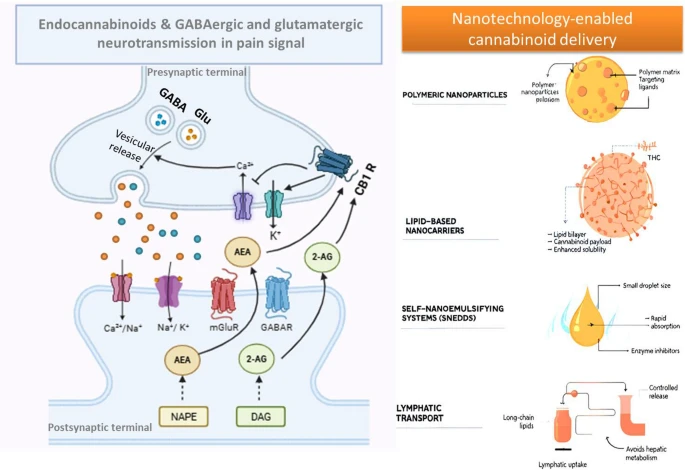
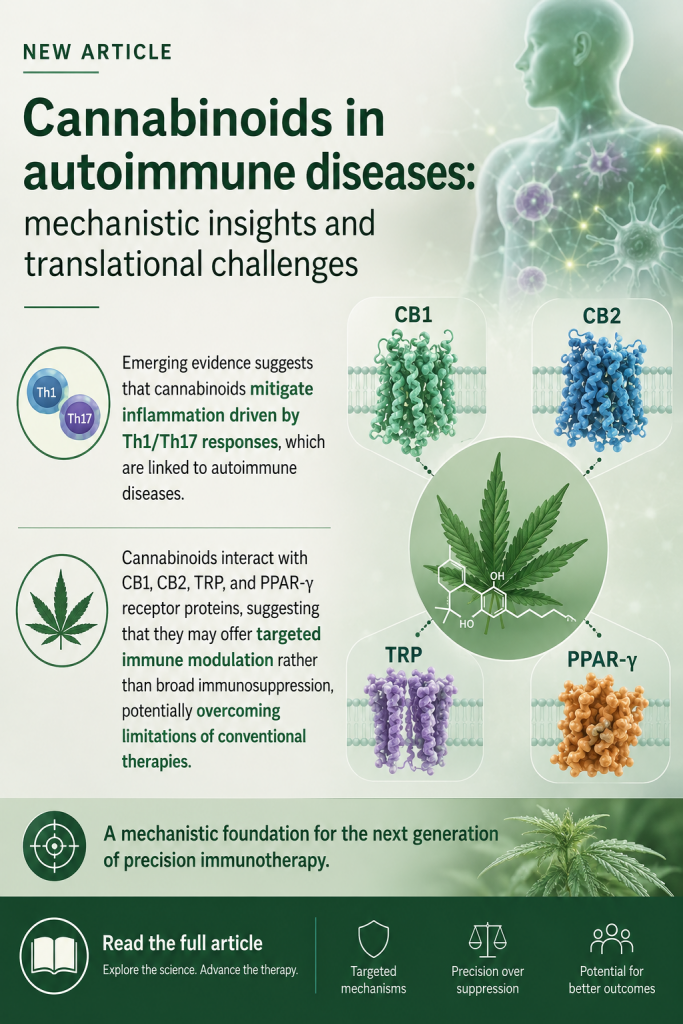
Preclinical and clinical evidence suggest that cannabinoids may modulate the endocannabinoid system, attenuate HPA axis hyperactivity, reduce neuroinflammation, and influence monoaminergic and neuroplasticity-related pathways.
This review synthesizes the current literature on the mechanistic links among the HPA axis, inflammation, and MDD, highlighting the emerging role of Cannabis sativa-derived compounds in targeting these interconnected pathways.”
https://pubmed.ncbi.nlm.nih.gov/42239513
https://www.frontiersin.org/journals/pharmacology/articles/10.3389/fphar.2026.1801474/full