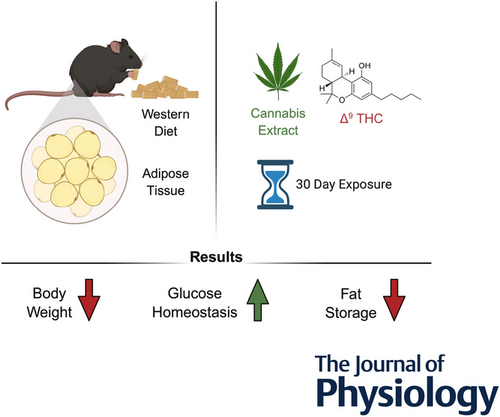
“Diet-induced obesity (DIO) is associated with dysregulated adipoinsular axis and endocannabinoid system (eCBS) function. Acute cannabis consumption stimulates appetite; however, chronic consumption is paradoxically associated with lower prevalence of human obesity and type 2 diabetes.
We investigated the impact of chronic exposure to Δ9 tetrahydrocannabinol (THC) and cannabis extracts on DIO and glucose homeostasis in mice.
Male mice were fed a high-fat/sucrose diet or a low-fat/no-sucrose diet for 60 days. At day 30, mice were administered THC (5 mg/kg) or cannabis extracts matched for THC content daily for 30 days. We assessed adipocyte biology, glucose tolerance, insulin sensitivity, eCBS expression, body weight, food intake and motor activity. Roles for the eCBS in cannabis-induced changes in metabolic processes, including cellular bioenergetics, were analysed in 3T3-L1 adipocytes.
THC and extracts reduced body weight and fat mass in DIO mice, and reversed DIO-associated changes in expression of adipokines that regulate the adipoinsular axis. Extracts normalized expression of adipokines more effectively than THC. Notably, extracts – but not THC – normalized glucose clearance in DIO mice to levels found in lean mice. In addition, THC and extracts promoted anti-adipogenic effects and changes in energy metabolism in 3T3-L1 cells in a concentration-dependent manner.
These studies suggest that chronic cannabinoid exposure improves metabolic function and dysregulated glucose homeostasis in DIO by a mechanism that includes restoring impaired adipoinsular axis function.
KEY POINTS: Δ9 Tetrahydrocannabinol (Δ9THC) and cannabis extracts reduce body weight and fat mass in obese mice. Cannabis extracts, but not Δ9THC alone, improve glucose homeostasis in obese mice. Extracts more effectively normalize expression of components of the adipoinsular axis in obese mice. Δ9THC and extracts promote anti-adipogenic effects in 3T3-L1 cells. Δ9THC and extracts alter cellular bioenergetics in 3T3-L1 cells.”
https://pubmed.ncbi.nlm.nih.gov/42113966
“In conclusion, this study demonstrates that chronic cannabinoid exposure, particularly with cannabis extract, reduces body weight, improves glucose homeostasis and normalizes adipose tissue function in a mouse model of DIO. Our findings highlight the potential therapeutic value of cannabinoids in managing obesity and related metabolic disorders, though further research is needed to fully understand the underlying mechanisms and translate these findings into clinical applications.”
https://physoc.onlinelibrary.wiley.com/doi/10.1113/JP290431