“The β-caryophyllene (BCP), a phytocannabinoid presents in various essential oils, demonstrated selective action on the CB2 endocannabinoid receptor and attracted considerable attention because of its several pharmacological activities. Despite this recognized potential, this hydrophobic compound is a volatile and acid-sensitive sesquiterpene that readily oxidizes when exposed to air, and has low bioavailability in oral formulations. Thus, the development of formulations that guarantee its stability and increase its bioavailability is a challenge for its use in the pharmaceutical field.
RESULTS:
The systems presented here may represent an interesting approach to overcome the limitations already mentioned for this terpene. These systems proved to be promising for improving solubility, stability and controlled release of this pharmacological relevant sesquiterpene. In the industrial field, some companies have filed patent applications for the commercial use of the BCP, however, the use of pharmaceutical formulations still appeared moderate.
CONCLUSION:
This prospective study evidenced the new perspectives related to BCP vectorization systems in the pharmaceutical and industrial marketing field and may serve as a basis for further research and pharmaceutical use of this powerful
cannabinoid.”
https://www.ncbi.nlm.nih.gov/pubmed/30207226
http://www.eurekaselect.com/165376/article
“β-caryophyllene (BCP) is a common constitute of the essential oils of numerous spice, food plants and major component in Cannabis.”
http://www.ncbi.nlm.nih.gov/pubmed/23138934]]>
 “Cannabinoid receptor 1 (CB1) is a promising therapeutic target for a variety of disorders. Distinct efficacy profiles showed different therapeutic effects on CB1 dependent on three classes of ligands: agonists, antagonists, and inverse agonists. To discriminate the distinct efficacy profiles of the ligands, we carried out molecular dynamics (MD) simulations to identify the dynamic behaviors of inactive and active conformations of CB1 structures with the ligands. In addition, the molecular mechanics Poisson-Boltzmann surface area (MM-PBSA) method was applied to analyze the binding free energy decompositions of the CB1-ligand complexes. With these two methods, we found the possibility that the three classes of ligands can be discriminated. Our findings shed light on the understanding of different efficacy profiles of ligands by analyzing the structural behaviors of intact CB1 structures and the binding energies of ligands, thereby yielding insights that are useful for the design of new potent CB1 drugs.”
https://www.ncbi.nlm.nih.gov/pubmed/30213978
https://www.nature.com/articles/s41598-018-31749-z
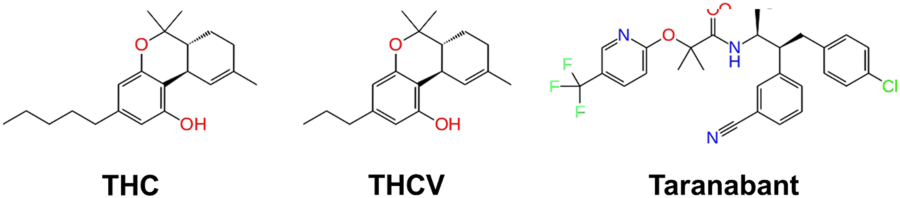
“Chemical structure of the partial agonist THC, antagonist THCV, and inverse agonist Taranabant.”
“Cannabinoid receptor 1 (CB1) is a promising therapeutic target for a variety of disorders. Distinct efficacy profiles showed different therapeutic effects on CB1 dependent on three classes of ligands: agonists, antagonists, and inverse agonists. To discriminate the distinct efficacy profiles of the ligands, we carried out molecular dynamics (MD) simulations to identify the dynamic behaviors of inactive and active conformations of CB1 structures with the ligands. In addition, the molecular mechanics Poisson-Boltzmann surface area (MM-PBSA) method was applied to analyze the binding free energy decompositions of the CB1-ligand complexes. With these two methods, we found the possibility that the three classes of ligands can be discriminated. Our findings shed light on the understanding of different efficacy profiles of ligands by analyzing the structural behaviors of intact CB1 structures and the binding energies of ligands, thereby yielding insights that are useful for the design of new potent CB1 drugs.”
https://www.ncbi.nlm.nih.gov/pubmed/30213978
https://www.nature.com/articles/s41598-018-31749-z
“Chemical structure of the partial agonist THC, antagonist THCV, and inverse agonist Taranabant.”

 “Drug addiction is a chronic relapsing disorder that produces a dramaticglobal health burden worldwide. Not effective treatment of drug addiction is currently available probably due to the difficulties to find an appropriate target to manage this complex disease raising the needs for further identification of novel therapeutic approaches.
The endocannabinoid system has been found to play a crucial role in the neurobiological substrate underlying drug addiction.
Endocannabinoids and cannabinoid receptors are widely expressed in the main areas of the mesocorticolimbic system that participate in the initiation and maintenance of drug consumption and in the development of compulsion and loss of behavioral control occurring during drug addiction.
The identification of the important role played by CB1 cannabinoid receptors in drug addiction encouraged the possible used of an early commercialized CB1 receptor antagonist for treating drug addiction.
However, the incidence of serious psychiatric adverse events leaded to the sudden withdrawal from the market of this CB1 antagonist and all the research programs developed by pharmaceutical companies to obtain new CB1 antagonists were stopped.
Currently, new research strategies are under development to target the endocannabinoid system for drug addiction avoiding these side effects, which include allosteric negative modulators of CB1 receptors and compounds targeting CB2 receptors.
Recent studies showing the potential role of CB2 receptors in the addictive properties of different drugs of abuse have open a promising research opportunity to develop novel possible therapeutic approaches.”
“Drug addiction is a chronic relapsing disorder that produces a dramaticglobal health burden worldwide. Not effective treatment of drug addiction is currently available probably due to the difficulties to find an appropriate target to manage this complex disease raising the needs for further identification of novel therapeutic approaches.
The endocannabinoid system has been found to play a crucial role in the neurobiological substrate underlying drug addiction.
Endocannabinoids and cannabinoid receptors are widely expressed in the main areas of the mesocorticolimbic system that participate in the initiation and maintenance of drug consumption and in the development of compulsion and loss of behavioral control occurring during drug addiction.
The identification of the important role played by CB1 cannabinoid receptors in drug addiction encouraged the possible used of an early commercialized CB1 receptor antagonist for treating drug addiction.
However, the incidence of serious psychiatric adverse events leaded to the sudden withdrawal from the market of this CB1 antagonist and all the research programs developed by pharmaceutical companies to obtain new CB1 antagonists were stopped.
Currently, new research strategies are under development to target the endocannabinoid system for drug addiction avoiding these side effects, which include allosteric negative modulators of CB1 receptors and compounds targeting CB2 receptors.
Recent studies showing the potential role of CB2 receptors in the addictive properties of different drugs of abuse have open a promising research opportunity to develop novel possible therapeutic approaches.”
 “The evolving policies regarding the use of therapeutic Cannabis have steadily increased the public interest in its use as a complementary and alternative medicine in several disorders, including inflammatory bowel disease.
Endocannabinoids represent both an appealing therapeutic strategy and a captivating scientific dilemma.
Results from clinical trials have to be carefully interpreted owing to possible reporting-biases related to cannabinoids psychotropic effects. Moreover, discriminating between symptomatic improvement and the real gain on the underlying inflammatory process is often challenging.
This review summarizes the advances and latest discovery in this ever-changing field of investigation, highlighting the main limitations in the current use of these drugs in clinical practice and the possible future perspectives to overcome these flaws.”
“The evolving policies regarding the use of therapeutic Cannabis have steadily increased the public interest in its use as a complementary and alternative medicine in several disorders, including inflammatory bowel disease.
Endocannabinoids represent both an appealing therapeutic strategy and a captivating scientific dilemma.
Results from clinical trials have to be carefully interpreted owing to possible reporting-biases related to cannabinoids psychotropic effects. Moreover, discriminating between symptomatic improvement and the real gain on the underlying inflammatory process is often challenging.
This review summarizes the advances and latest discovery in this ever-changing field of investigation, highlighting the main limitations in the current use of these drugs in clinical practice and the possible future perspectives to overcome these flaws.”
 “The chronic use of drugs that reduce the dopaminergic neurotransmission can cause a hyperkinetic movement disorder called tardive dyskinesia (TD). The pathophysiology of this disorder is not entirely understood but could involve oxidative and neuroinflammatory mechanisms.
“The chronic use of drugs that reduce the dopaminergic neurotransmission can cause a hyperkinetic movement disorder called tardive dyskinesia (TD). The pathophysiology of this disorder is not entirely understood but could involve oxidative and neuroinflammatory mechanisms.
 “Cannabis sativa contains many related compounds known as phytocannabinoids. The main psychoactive and non-psychoactive compounds are Δ9-tetrahydrocannabidiol (THC) and
“Cannabis sativa contains many related compounds known as phytocannabinoids. The main psychoactive and non-psychoactive compounds are Δ9-tetrahydrocannabidiol (THC) and 