 “The Cannabis plant has been used for many of years as a medicinal agent in the relief of pain and seizures. It contains approximately 540 natural compounds including more than 100 that have been identified as phytocannabinoids due to their shared chemical structure. The predominant psychotropic component is Δ9-tetrahydrocannabinol (Δ9-THC), while the major non-psychoactive ingredient is cannabidiol (CBD). These compounds have been shown to be partial agonists or antagonists at the prototypical cannabinoid receptors, CB1 and CB2. The therapeutic actions of Δ9-THC and CBD include an ability to act as analgesics, anti-emetics, anti-inflammatory agents, anti-seizure compounds and as protective agents in neurodegeneration. However, there is a lack of well-controlled, double blind, randomized clinical trials to provide clarity on the efficacy of either Δ9-THC or CBD as therapeutics. Moreover, the safety concerns regarding the unwanted side effects of Δ9-THC as a psychoactive agent preclude its widespread use in the clinic. The legalization of cannabis for medicinal purposes and for recreational use in some regions will allow for much needed research on the pharmacokinetics and pharmocology of medical cannabis. This brief review focuses on the use of cannabis as a medicinal agent in the treatment of pain, epilepsy and neurodegenerative diseases. Despite the paucity of information, attention is paid to the mechanisms by which medical cannabis may act to relieve pain and seizures.”
“The Cannabis plant has been used for many of years as a medicinal agent in the relief of pain and seizures. It contains approximately 540 natural compounds including more than 100 that have been identified as phytocannabinoids due to their shared chemical structure. The predominant psychotropic component is Δ9-tetrahydrocannabinol (Δ9-THC), while the major non-psychoactive ingredient is cannabidiol (CBD). These compounds have been shown to be partial agonists or antagonists at the prototypical cannabinoid receptors, CB1 and CB2. The therapeutic actions of Δ9-THC and CBD include an ability to act as analgesics, anti-emetics, anti-inflammatory agents, anti-seizure compounds and as protective agents in neurodegeneration. However, there is a lack of well-controlled, double blind, randomized clinical trials to provide clarity on the efficacy of either Δ9-THC or CBD as therapeutics. Moreover, the safety concerns regarding the unwanted side effects of Δ9-THC as a psychoactive agent preclude its widespread use in the clinic. The legalization of cannabis for medicinal purposes and for recreational use in some regions will allow for much needed research on the pharmacokinetics and pharmocology of medical cannabis. This brief review focuses on the use of cannabis as a medicinal agent in the treatment of pain, epilepsy and neurodegenerative diseases. Despite the paucity of information, attention is paid to the mechanisms by which medical cannabis may act to relieve pain and seizures.”
https://www.ncbi.nlm.nih.gov/pubmed/31332738
https://link.springer.com/chapter/10.1007%2F978-3-030-21737-2_8

 “Delta-9-tetrahydrocannabinol (THC) is the primary psychoactive compound in
“Delta-9-tetrahydrocannabinol (THC) is the primary psychoactive compound in 
 “Herein, we report the antioxidant activity of
“Herein, we report the antioxidant activity of 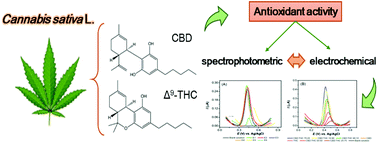
 “The purpose of the study was to describe associations between employment and marijuana use among adolescents 2 years before passage of 2012 ballot initiative and 2 years after the implementation of retail recreational marijuana sales took place in Washington.
“The purpose of the study was to describe associations between employment and marijuana use among adolescents 2 years before passage of 2012 ballot initiative and 2 years after the implementation of retail recreational marijuana sales took place in Washington.
