
There is some evidence that cannabidiol (CBD) can induce cell death and increases the radiosensitivity of GBM by enhancing apoptosis. Beside initiation of death, CBD has been demonstrated as an inducer of autophagy.
In the present study, we address the question whether CBD simultaneously induces a protective effect in GBM by upregulating autophagy. Addition of chloroquine that suppressed autophagic flux to 2D GBM cultures increased CBD-induced cell death, presenting proof for the protective autophagy.
Blockage of autophagy upregulated radiation-induced cytotoxicity but only modestly affected the levels of cell death in CBD- or CBD/γ-irradiated 3D GBM cultures. Furthermore, CBD enhanced the pro-apoptotic activities of JNK1/2 and MAPK p38 signaling cascades while partially downregulated the pro-survival PI3K-AKT cascade, thereby changing a balance between cell death and survival.
Suppression of JNK activation partially reduced CBD-induced cell death in 3D GBM cultures. In contrast, co-treatment of CBD-targeted cells with inhibitors of PI3K-AKT-NF-κB, IKK-NF-κB or JAK2-STAT3 pathways killed surviving GBM cells in both 2D and 3D cultures, potentially improving the therapeutic ratio of GBM.”
https://www.ncbi.nlm.nih.gov/pubmed/32060308
“Killing efficiency of cannabinoids (CBD, THC and their combination CBD+THC) against GBM in vitro and in animal experiments has been elucidated in numerous studies during the last 15 years. Additional investigations also confirmed a cytotoxic role of cannabinoids for several other types of cancer. A number of studies demonstrated the efficiency of combined treatments of cannabinoids together with γ-irradiation in both cell culture and in animal experiments.”

 “Patients with non-small cell lung cancer (NSCLC) develop resistance to antitumor agents by mechanisms that involve the epithelial-to-mesenchymal transition (EMT). This necessitates the development of new complementary drugs, e.g.,
“Patients with non-small cell lung cancer (NSCLC) develop resistance to antitumor agents by mechanisms that involve the epithelial-to-mesenchymal transition (EMT). This necessitates the development of new complementary drugs, e.g.,  “Novel anticancer medicines, including targeted therapies and immune checkpoint inhibitors, have greatly improved the management of cancers. However, both conventional and new anticancer treatments induce cardiac adverse effects, which remain a critical issue in clinic.
“Novel anticancer medicines, including targeted therapies and immune checkpoint inhibitors, have greatly improved the management of cancers. However, both conventional and new anticancer treatments induce cardiac adverse effects, which remain a critical issue in clinic. “Cannabinoids are a group of structurally heterogeneous but pharmacologically related compounds, including plant-derived cannabinoids, synthetic substances and endogenous cannabinoids, such as anandamide and 2-arachidonoylglycerol.
“Cannabinoids are a group of structurally heterogeneous but pharmacologically related compounds, including plant-derived cannabinoids, synthetic substances and endogenous cannabinoids, such as anandamide and 2-arachidonoylglycerol. “In the last decade, we have observed an increased public and scientific interest in the clinical applications of medical cannabis.
“In the last decade, we have observed an increased public and scientific interest in the clinical applications of medical cannabis. “Endo-, phyto- and synthetic cannabinoids have been proposed as promising anti-cancer agents able to impair cancer cells’ behavior without affecting their non-transformed counterparts.
“Endo-, phyto- and synthetic cannabinoids have been proposed as promising anti-cancer agents able to impair cancer cells’ behavior without affecting their non-transformed counterparts.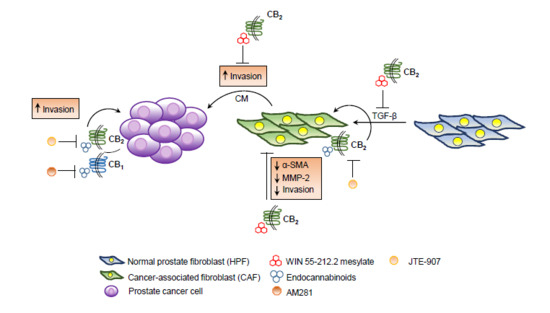

 “Recent changes to the legal status of marijuana in Canada warrant a review of the information that patients and families are accessing online regarding the role of
“Recent changes to the legal status of marijuana in Canada warrant a review of the information that patients and families are accessing online regarding the role of  “The aim of this review article is to summarize current knowledge about the role of cannabinoids and cannabinoid receptors in tumor disease modulation and to evaluate comprehensively the use of cannabinoids in cancer patients.
“The aim of this review article is to summarize current knowledge about the role of cannabinoids and cannabinoid receptors in tumor disease modulation and to evaluate comprehensively the use of cannabinoids in cancer patients. “Tetrahydrocannabinol (THC), cannabidiol (CBD) and cannabinol (CBN) affect the human endocannabinoid system.
“Tetrahydrocannabinol (THC), cannabidiol (CBD) and cannabinol (CBN) affect the human endocannabinoid system.