 “Cannabinoids such as ▵-9-THC and CBD can downregulate the immune response by modulating the endocannabinoid system. This modulation is relevant for the treatment of prevalent autoimmune diseases (ADs), such as multiple sclerosis (MS), systemic lupus erythematosus (SLE), diabetes mellitus type 1 (DMT1), and rheumatoid arthritis (RA). These conditions require new therapeutic options with fewer side effects for the control of the autoimmune response. Objective: to conduct a literature review of preclinical scientific evidence that supports further clinical investigations for the use of cannabinoids (natural or synthetic) as potential immunomodulators of the immune response in ADs.
“Cannabinoids such as ▵-9-THC and CBD can downregulate the immune response by modulating the endocannabinoid system. This modulation is relevant for the treatment of prevalent autoimmune diseases (ADs), such as multiple sclerosis (MS), systemic lupus erythematosus (SLE), diabetes mellitus type 1 (DMT1), and rheumatoid arthritis (RA). These conditions require new therapeutic options with fewer side effects for the control of the autoimmune response. Objective: to conduct a literature review of preclinical scientific evidence that supports further clinical investigations for the use of cannabinoids (natural or synthetic) as potential immunomodulators of the immune response in ADs.
Methodology: A systematic search was carried out in different databases using different MeSH terms, such as Cannabis sativa L., cannabinoids, immunomodulation, and ADs. Initially, 677 journal articles were found. After filtering by publication date (from 2000 to 2020 for SLE, DMT1, and RA; and 2010 to 2020 for MS) and removing the duplicate items, 200 articles were selected and analyzed by title and summary associated with the use of cannabinoids as immunomodulatory treatment for those diseases.
Results: Evidence of the immunomodulatory effect of cannabinoids in the diseases previously mentioned, but SLE that did not meet the search criteria, was summarized from 24 journal articles. CBD was found to be one of the main modulators of the immune response. This molecule decreased the number of Th1 and Th17 proinflammatory cells and the production of the proinflammatory cytokines, interleukin (IL)-1, IL-12, IL-17, interferon (IFN)-γ, and tumor necrosis factor alpha, in mouse models of MS and DMT1. Additionally, new synthetic cannabinoid-like molecules, with agonist or antagonist activity on CB1, CB2, TRPV1, PPAR-α, and PPAR-γ receptors, have shown anti-inflammatory properties in MS, DMT1, and RA.
Conclusion: Data from experimental animal models of AD showed that natural and synthetic cannabinoids downregulate inflammatory responses mediated by immune cells responsible for AD chronicity and progression. Although synthetic cannabinoid-like molecules were evaluated in just two clinical trials, they corroborated the potential use of cannabinoids to treat some ADs. Notwithstanding, new cannabinoid-based approaches are required to provide alternative treatments to patients affected by the large group of ADs.”
https://pubmed.ncbi.nlm.nih.gov/34030476/
https://www.liebertpub.com/doi/10.1089/can.2020.0183

 “Agents targeting the endocannabinoid system (ECS) have gained attention as potential cancer treatments. Given recent evidence that cannabinoid receptor 2 (CB2R) regulates lymphocyte development and inflammation, we performed studies on CB2R in the immune response against melanoma. Analysis of The Cancer Genome Atlas (TCGA) data revealed a strong positive correlation between CB2R expression and survival, as well as B cell infiltration in human melanoma. In a murine melanoma model, CB2R expression reduced the growth of melanoma as well as the B cell frequencies in the tumor microenvironment (TME), compared to CB2R-deficient mice. In depth analysis of tumor-infiltrating B cells using single-cell RNA sequencing suggested a less differentiated phenotype in tumors from
“Agents targeting the endocannabinoid system (ECS) have gained attention as potential cancer treatments. Given recent evidence that cannabinoid receptor 2 (CB2R) regulates lymphocyte development and inflammation, we performed studies on CB2R in the immune response against melanoma. Analysis of The Cancer Genome Atlas (TCGA) data revealed a strong positive correlation between CB2R expression and survival, as well as B cell infiltration in human melanoma. In a murine melanoma model, CB2R expression reduced the growth of melanoma as well as the B cell frequencies in the tumor microenvironment (TME), compared to CB2R-deficient mice. In depth analysis of tumor-infiltrating B cells using single-cell RNA sequencing suggested a less differentiated phenotype in tumors from 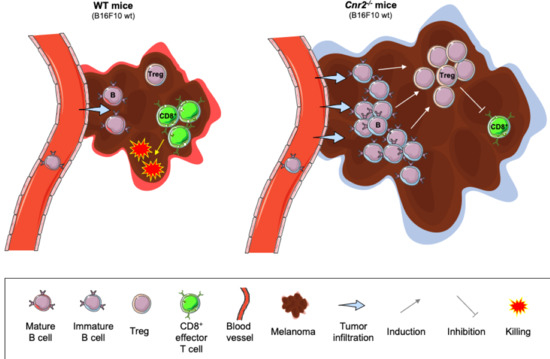

 “Cannabis was extensively utilized for its medicinal properties till the 19th century. A steep decline in its medicinal usage was observed later due to its emergence as an illegal recreational drug. Advances in technology and scientific findings led to the discovery of delta-9-tetrahydrocannabinol (THC), the primary psychoactive compound of cannabis, that further led to the discovery of endogenous cannabinoids system consisting of G-protein-coupled receptors – cannabinoid receptor 1 and cannabinoid receptor 2 along with their ligands, mainly anandamide and 2-arachidonoylglycerol. Endocannabinoid (EC) is shown to be a modulator not only for physiological functions but also for the immune system, endocrine network, and central nervous system. Medicinal research and meta-data analysis over the last few decades have shown a significant potential for both THC and cannabidiol (CBD) to exert palliative effects. People suffering from many forms of advanced stages of cancers undergo chemotherapy-induced nausea and vomiting followed by severe and chronic neuropathic pain and weight loss. THC and CBD exhibit effective analgesic, anxiolytic, and appetite-stimulating effect on patients suffering from cancer. Drugs currently available in the market to treat such chemotherapy-induced cancer-related ailments are Sativex (GW Pharmaceutical), Dronabinol (Unimed Pharmaceuticals), and Nabilone (Valeant Pharmaceuticals). Apart from exerting palliative effects, THC also shows promising role in the treatment of cancer growth, neurodegenerative diseases (multiple sclerosis and Alzheimer’s disease), and alcohol addiction and hence should be exploited for potential benefits. The current review discusses the nature and role of CB receptors, specific applications of cannabinoids, and major studies that have assessed the role of cannabinoids in cancer management.”
“Cannabis was extensively utilized for its medicinal properties till the 19th century. A steep decline in its medicinal usage was observed later due to its emergence as an illegal recreational drug. Advances in technology and scientific findings led to the discovery of delta-9-tetrahydrocannabinol (THC), the primary psychoactive compound of cannabis, that further led to the discovery of endogenous cannabinoids system consisting of G-protein-coupled receptors – cannabinoid receptor 1 and cannabinoid receptor 2 along with their ligands, mainly anandamide and 2-arachidonoylglycerol. Endocannabinoid (EC) is shown to be a modulator not only for physiological functions but also for the immune system, endocrine network, and central nervous system. Medicinal research and meta-data analysis over the last few decades have shown a significant potential for both THC and cannabidiol (CBD) to exert palliative effects. People suffering from many forms of advanced stages of cancers undergo chemotherapy-induced nausea and vomiting followed by severe and chronic neuropathic pain and weight loss. THC and CBD exhibit effective analgesic, anxiolytic, and appetite-stimulating effect on patients suffering from cancer. Drugs currently available in the market to treat such chemotherapy-induced cancer-related ailments are Sativex (GW Pharmaceutical), Dronabinol (Unimed Pharmaceuticals), and Nabilone (Valeant Pharmaceuticals). Apart from exerting palliative effects, THC also shows promising role in the treatment of cancer growth, neurodegenerative diseases (multiple sclerosis and Alzheimer’s disease), and alcohol addiction and hence should be exploited for potential benefits. The current review discusses the nature and role of CB receptors, specific applications of cannabinoids, and major studies that have assessed the role of cannabinoids in cancer management.” “Recently, there has been a growing interest in the medical applications of Cannabis plants. They owe their unique properties to a group of secondary metabolites known as phytocannabinoids, which are specific for this genus. Phytocannabinoids, and cannabinoids generally, can interact with cannabinoid receptors being part of the endocannabinoid system present in animals. Over the years a growing body of scientific evidence has been gathered, suggesting that these compounds have therapeutic potential.
“Recently, there has been a growing interest in the medical applications of Cannabis plants. They owe their unique properties to a group of secondary metabolites known as phytocannabinoids, which are specific for this genus. Phytocannabinoids, and cannabinoids generally, can interact with cannabinoid receptors being part of the endocannabinoid system present in animals. Over the years a growing body of scientific evidence has been gathered, suggesting that these compounds have therapeutic potential.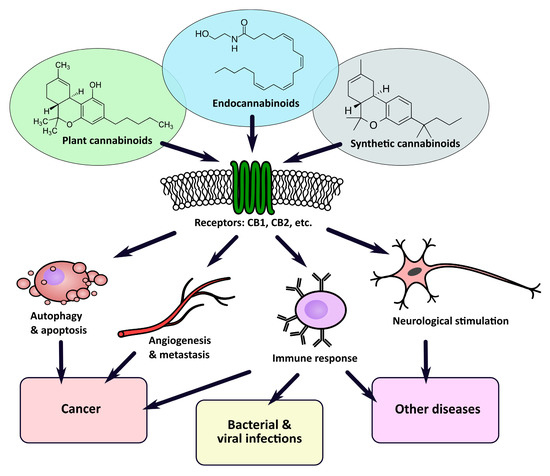
 “Plant-based therapies date back centuries. Cannabis sativa is one such plant that was used medicinally up until the early part of the 20th century.
“Plant-based therapies date back centuries. Cannabis sativa is one such plant that was used medicinally up until the early part of the 20th century.
 “Autism spectrum disorder (ASD) is a complex neurodevelopmental disorder with a multifactorial etiology. Latest researches are raising the hypothesis of a link between the onset of the main behavioral symptoms of ASD and the chronic neuroinflammatory condition of the autistic brain; increasing evidence of this connection is shedding light on new possible players in the pathogenesis of ASD.
“Autism spectrum disorder (ASD) is a complex neurodevelopmental disorder with a multifactorial etiology. Latest researches are raising the hypothesis of a link between the onset of the main behavioral symptoms of ASD and the chronic neuroinflammatory condition of the autistic brain; increasing evidence of this connection is shedding light on new possible players in the pathogenesis of ASD.