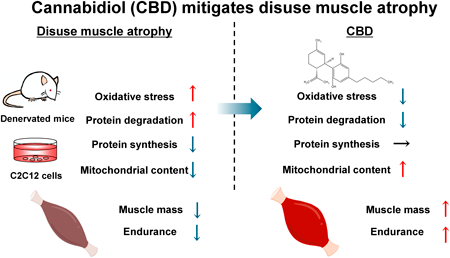
“In the experiment, we tested the effect of 30% CBD oil on the activity of the antioxidant system (superoxide dismutase, catalase, glutathione peroxidase, glutathione), the level of total antioxidant capacity, and the concentrations of ions (calcium, magnesium, and phosphorus) in honeybee workers in the hive test.
For this purpose, we prepared hives containing all stages of the development of honey bees and started the experiment by adding 200 marked, one-day old bees to each colony (intended for hemolymph collection). In the test, we created three groups (two colonies per group): (1) Experimental with CBD oil mixed with sugar syrup (CSy); (2) experimental with CBD oil on textile strips (CSt); and (3) control with pure sugar syrup only (C). Every week, we collected hemolymph from the marked bees.
In the experiment, all antioxidant enzyme activities were higher for the experimental groups CSy and CSt compared to group C. The highest concentrations/levels were obtained for the CSy group. Concentrations of calcium, magnesium, and phosphorus ions were also higher for the experimental groups compared to the C group (the highest concentration for the CSy group).
We conclude that CBD oil positively contributes to stimulating the antioxidant system of honeybees.”
https://pubmed.ncbi.nlm.nih.gov/36829838
“Our previous studies have shown that CBD oil/hemp extracts added to the diet of bees prolonged the life of insects and contributed to a positive stimulation of the immune system”
“The aim of this study was to evaluate the effect of a commercial hemp extract in the form of CBD oil on the activity of the antioxidant system”
“After a series of studies in cages and in colony conditions, we found that supplementation with CBD will potentially support the immune system of honeybees through stimulating the antioxidant system (protection against oxidative stress affecting cells and their biochemistry).
Depending on the need, the effects can be obtained regardless of the method of administration, but for the best results, we suggest using CBD in nutritional supplements (direct, faster action). In addition, research confirms that the active substance CBD may be responsible for the positive effect of the hemp extract.”