




 “∆ 9 -Tetrahydrocannabinol (∆9 -THC), the active phytocannabinoid in cannabis, is virtually an adjunct to the endogenous endocannabinoid signaling system.
“∆ 9 -Tetrahydrocannabinol (∆9 -THC), the active phytocannabinoid in cannabis, is virtually an adjunct to the endogenous endocannabinoid signaling system.
By interacting with G-protein-coupled receptors CB1 and CB2, ∆9 -THC affects peripheral and central circulation by lowering sympathetic activity, altering gene expression, cell proliferation, and differentiation, decreasing leukocyte migration, modulating neurotransmitter release thereby modulating cardiovascular functioning, tumorigenesis, immune responses, behavioral and locomotory activities respectively.
∆ 9 -THC is effective in suppressing chemotherapy-induced vomiting, retards malignant tumor growth, inhibits metastasis, and promotes apoptosis. Other mechanisms involved are targeting cell cycle at the G2-M phase in human breast cancer, downregulation of E2F transcription factor 1 (E2F1) in human glioblastoma multiforme, and stimulation of ER stress-induced autophagy.
∆ 9 -THC also plays a role in ameliorating neuroinflammation, excitotoxicity, neuroplasticity, trauma, and stroke and is associated with reliving childhood epilepsy, brain trauma, and neurodegenerative diseases.
∆9 -THC via CB1 receptors affects nociception, emotion, memory, and reduces neuronal excitability and excitotoxicity in epilepsy. It also increases renal blood flow, reduces intraocular pressure via a sympathetic pathway, and modulates hormonal release, thereby decreasing the reproductive function and increasing glucose metabolism.
Versatile medical marijuana has stimulated abundant research demonstrating substantial therapeutic promise, suggesting the possibilities of first-in-class drugs in diverse therapeutic segments. In this review, we represent the current pharmacological status of the phytocannabinoid, ∆ 9 -THC, and synthetic analogs in cancer, cardiovascular, and neurodegenerative disorders.”
 “While activation of cannabinoid (CB2) receptors has been shown to be neuroprotective, no studies have examined whether this neuroprotection is directed at cerebral arterioles and no studies have examined whether activation of CB2 receptors can rescue cerebrovascular dysfunction during a chronic disease state such as type 1 diabetes (T1D).
“While activation of cannabinoid (CB2) receptors has been shown to be neuroprotective, no studies have examined whether this neuroprotection is directed at cerebral arterioles and no studies have examined whether activation of CB2 receptors can rescue cerebrovascular dysfunction during a chronic disease state such as type 1 diabetes (T1D).
Our goal was to test the hypothesis that administration of a CB2 agonist (JWH-133) would improve impaired endothelial (eNOS)- and neuronal (nNOS)- dependent dilation of cerebral arterioles during T1D.
In vivo diameter of cerebral arterioles in nondiabetic and T1D rats was measured in response to an eNOS-dependent agonist (adenosine 5′-diphosphate; ADP), an nNOS-dependent agonist (N-methyl-D-aspartate; NMDA), and an NOS-independent agonist (nitroglycerin) before and 1 hour following JWH-133 (1 mg/kg IP). Dilation of cerebral arterioles to ADP and NMDA was greater in nondiabetic than in T1D rats.
Treatment with JWH-133 increased responses of cerebral arterioles to ADP and NMDA in both nondiabetic and T1D rats. Responses of cerebral arterioles to nitroglycerin were similar between nondiabetic and T1D rats, and JWH-133 did not influence responses to nitroglycerin in either group. The restoration in responses to the agonists by JWH-133 could be inhibited by treatment with a specific inhibitor of CB2 receptors (AM-630; 3 mg/kg IP).
Thus, activation of CB2 receptors can potentiate reactivity of cerebral arterioles during physiologic and pathophysiologic states. We speculate that treatment with CB2 receptor agonists may have potential therapeutic benefits for the treatment of cerebral vascular diseases via a mechanism that can increase cerebral blood flow.”
https://pubmed.ncbi.nlm.nih.gov/32979391/
“Activation of CB2 receptors improves cerebral vascular function. Activation of CB2 receptors improves responses in type 1 diabetes. We speculate that treatment with CB2 receptor agonists may have potential therapeutic benefits for the treatment of cerebral vascular disease that can contribute to the pathogenesis of stroke.”
https://www.sciencedirect.com/science/article/pii/S0026286220301370?via%3Dihub
 “Non-Uremic Calciphylaxis (NUC) is a rare condition that often manifests as intractable and painful integumentary wounds, afflicting patients with a high burden of co-morbidity.
“Non-Uremic Calciphylaxis (NUC) is a rare condition that often manifests as intractable and painful integumentary wounds, afflicting patients with a high burden of co-morbidity.
The Endocannabinoid System (ECS) is a ubiquitous signalling system that is theorised to be dysregulated within wound beds and associated peri-wound tissues.
Preclinical research has shown that the dominant chemical classes derived from the cannabis plant, cannabinoids, terpenes, and flavonoids, interact with the integumentary ECS to promote wound closure and analgesia.
This is a prospective open label cohort study involving two elderly Caucasian females with recalcitrant NUC leg ulcers of greater than 6 months duration.
Topical Cannabis-Based Medicines (TCBM) composed of cannabinoids, terpenes, and flavonoids were applied daily to both the wound bed and peri-wound tissues until complete wound closure was achieved.
Wounds were photographed regularly, and the digital images were subjected to planimetric analysis to objectively quantify the degree of granulation and epithelization. Analgesic utilisation, as a surrogate/proxy for pain scores, was also tracked. The cohort had a mean M3 multimorbidity index score of 3.31. Complete wound closure was achieved in a mean of 76.3 days. Additionally, no analgesics were required after a mean of 63 days.
The treatments were well tolerated with no adverse reactions. The positive results demonstrated in very challenging wounds such as NUC, among highly complex patients, suggest that TCBM may have an even broader role within integumentary and wound management.
This treatment paradigm warrants being trialled in other wound types and classes, and ultimately should be subjected to randomised controlled trials.”
https://pubmed.ncbi.nlm.nih.gov/32875692/
“Topical Cannabis‐Based Medicines, applied to both wound beds and peri‐wound tissues, represent a promising novel, non‐invasive, and safe treatment option for NUC leg ulcers.”
https://onlinelibrary.wiley.com/doi/full/10.1111/iwj.13484

 “There is no argument with regard to the physical and psychological stress-related nature of neuropsychiatric disorders. Yet, the mechanisms that facilitate disease onset starting from molecular stress responses are elusive.
“There is no argument with regard to the physical and psychological stress-related nature of neuropsychiatric disorders. Yet, the mechanisms that facilitate disease onset starting from molecular stress responses are elusive.
Environmental stress challenges individuals’ equilibrium, enhancing homeostatic request in the attempt to steer down arousal-instrumental molecular pathways that underlie hypervigilance and anxiety.
A relevant homeostatic pathway is the endocannabinoid system (ECS).
In this review, we summarize recent discoveries unambiguously listing ECS as a stress coping mechanism.
As stress evokes huge excitatory responses in emotional-relevant limbic areas, the ECS limits glutamate release via 2-arachydonilglycerol (2-AG) stress-induced synthesis and retrograde cannabinoid 1 (CB1)-receptor activation at the synapse. However, ECS shows intrinsic vulnerability as 2-AG overstimulation by chronic stress rapidly leads to CB1-receptor desensitization.
In this review, we emphasize the protective role of 2-AG in stress-response termination and stress resiliency. Interestingly, we discuss ECS regulation with a further nuclear homeostatic system whose nature is exquisitely epigenetic, orchestrated by Lysine Specific Demethylase 1.
We here emphasize a remarkable example of stress-coping network where transcriptional homeostasis subserves synaptic and behavioral adaptation, aiming at reducing psychiatric effects of traumatic experiences.”
https://pubmed.ncbi.nlm.nih.gov/32872402/
https://www.mdpi.com/1422-0067/21/17/6252

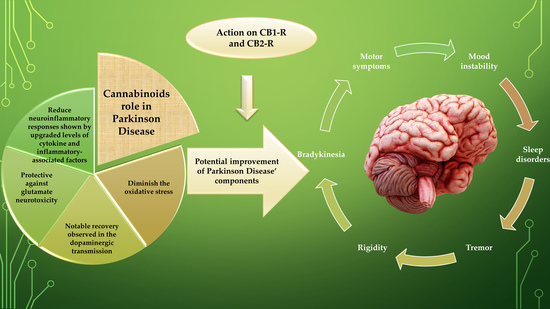
 “Current pharmacotherapy of Parkinson’s disease (PD) is symptomatic and palliative, with levodopa/carbidopa therapy remaining the prime treatment, and nevertheless, being unable to modulate the progression of the neurodegeneration. No available treatment for PD can enhance the patient’s life-quality by regressing this diseased state.
“Current pharmacotherapy of Parkinson’s disease (PD) is symptomatic and palliative, with levodopa/carbidopa therapy remaining the prime treatment, and nevertheless, being unable to modulate the progression of the neurodegeneration. No available treatment for PD can enhance the patient’s life-quality by regressing this diseased state.
Various studies have encouraged the enrichment of treatment possibilities by discovering the association of the effects of the endocannabinoid system (ECS) in PD.
These reviews delineate the reported evidence from the literature on the neuromodulatory role of the endocannabinoid system and expression of cannabinoid receptors in symptomatology, cause, and treatment of PD progression, wherein cannabinoid (CB) signalling experiences alterations of biphasic pattern during PD progression.
Endocannabinoids regulate the basal ganglia neuronal circuit pathways, synaptic plasticity, and motor functions via communication with dopaminergic, glutamatergic, and GABAergic signalling systems bidirectionally in PD.
Further, gripping preclinical and clinical studies demonstrate the context regarding the cannabinoid compounds, which is supported by various evidence (neuroprotection, suppression of excitotoxicity, oxidative stress, glial activation, and additional benefits) provided by cannabinoid-like compounds (much research addresses the direct regulation of cannabinoids with dopamine transmission and other signalling pathways in PD).
More data related to endocannabinoids efficacy, safety, and pharmacokinetic profiles need to be explored, providing better insights into their potential to ameliorate or even regress PD.”
https://pubmed.ncbi.nlm.nih.gov/32872273/
https://www.mdpi.com/1422-0067/21/17/6235
 “The endocannabinoid signaling system (ECSS) is altered by exposure to stress and mediates and modulates the effects of stress on the brain.
“The endocannabinoid signaling system (ECSS) is altered by exposure to stress and mediates and modulates the effects of stress on the brain.
Considerable preclinical data support critical roles for the endocannabinoids and their target, the CB1 cannabinoid receptor, in the adaptation of the brain to repeated stress exposure.
Chronic stress exposure increases vulnerability to mental illness, so the ECSS has attracted attention as a potential therapeutic target for the prevention and treatment of stress-related psychopathology.
We discuss human genetic studies indicating that the ECSS contributes to risk for mental illness in those exposed to severe stress and trauma early in life, and we explore the potential difficulties in pharmacological manipulation of the ECSS.”
“Inspired by the medicinal properties of the plant Cannabis sativa and its principal component (-)-trans-Δ9-tetrahydrocannabinol (THC), researchers have developed a variety of compounds to modulate the endocannabinoid system in the human brain.
Inhibitors of fatty acid amide hydrolase (FAAH) and monoacylglycerol lipase (MAGL), which are the enzymes responsible for the inactivation of the endogenous cannabinoids anandamide and 2-arachidonoylglycerol, respectively, may exert therapeutic effects without inducing the adverse side effects associated with direct cannabinoid CB1 receptor stimulation by THC.
Here we review the FAAH and MAGL inhibitors that have reached clinical trials, discuss potential caveats, and provide an outlook on where the field is headed.”
https://pubmed.ncbi.nlm.nih.gov/32867595/
https://www.annualreviews.org/doi/10.1146/annurev-pharmtox-030220-112741

Body: Both ACE inhibitors and ARBs induce overexpression of the angiotensin converting enzyme 2 (ACE-2) receptor, which has been identified as the main receptor used by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) to enter into the alveolar cells of the lungs. While cannabinoids are known to reduce hypertension, the studies testing the hypotensive effects of cannabinoids never addressed their effects on ACE-2 receptors. However, some studies have linked the endocannabinoid system (ECS) with the renin angiotensin system (RAS), including a cross-modulation between the cannabinoid receptor 1 (CB1) and angiotensin II levels.
Conclusion: Since there are around 192 million people using cannabis worldwide, we believe that the mechanism underlying the hypotensive properties of cannabinoids should be urgently studied to understand if they can also lead to ACE-2 overexpression as other antihypertensive drugs do.”
https://pubmed.ncbi.nlm.nih.gov/32835160/
https://jcannabisresearch.biomedcentral.com/articles/10.1186/s42238-020-00030-4
 “Alzheimer’s disease (AD) is an irreversible chronic neurodegenerative disorder that occurs when neurons in the brain degenerate and die. Pain frequently arises in older patients with neurodegenerative diseases including AD. However, the presence of pain in older people is usually overlooked with cognitive dysfunctions. Most of the times dementia patients experience moderate to severe pain but the development of severe cognitive dysfunctions tremendously affects their capability to express the presence of pain. Currently, there are no effective treatments against AD that emphasize the necessity for increasing research to develop novel drugs for treating or preventing the disease process. Furthermore, the prospective therapeutic use of cannabinoids in AD has been studied for the past few years. In this regard, targeting the endocannabinoid system has considered as a probable therapeutic strategy to control several associated pathological pathways, such as mitochondrial dysfunction, excitotoxicity, oxidative stress, and neuroinflammation for the management of AD. In this review, we focus on recent studies about the role of cannabinoids for the treatment of pain and related neuropathological changes in AD.”
“Alzheimer’s disease (AD) is an irreversible chronic neurodegenerative disorder that occurs when neurons in the brain degenerate and die. Pain frequently arises in older patients with neurodegenerative diseases including AD. However, the presence of pain in older people is usually overlooked with cognitive dysfunctions. Most of the times dementia patients experience moderate to severe pain but the development of severe cognitive dysfunctions tremendously affects their capability to express the presence of pain. Currently, there are no effective treatments against AD that emphasize the necessity for increasing research to develop novel drugs for treating or preventing the disease process. Furthermore, the prospective therapeutic use of cannabinoids in AD has been studied for the past few years. In this regard, targeting the endocannabinoid system has considered as a probable therapeutic strategy to control several associated pathological pathways, such as mitochondrial dysfunction, excitotoxicity, oxidative stress, and neuroinflammation for the management of AD. In this review, we focus on recent studies about the role of cannabinoids for the treatment of pain and related neuropathological changes in AD.”
https://pubmed.ncbi.nlm.nih.gov/32792944/
“Cannabinoids act by targeting several signaling processes, such as pain, abnormal processing of Aβ and tau, neuroinflammation, excitotoxicity, oxidative stress, and mitochondrial dysfunction, which play a pivotal role in the management of AD. Cannabinoids also ameliorate behavioral and cognitive dysfunctions. Therefore, due to these extensive medical uses of cannabinoid compounds, it can be said that targeting the endocannabinoid system can be a promising strategy to develop an effective therapy for the management of AD. Furthermore, cannabinoids may demonstrate a safe and reliable low-cost therapy, with limited side effects.”
https://www.frontiersin.org/articles/10.3389/fphar.2020.01097/full