“INTRODUCTION
: The increased incidence of Glioblastoma Multiforme, the most aggressive and most common primary brain tumour, is evident worldwide. Survival rates are reaching only 15 months due to its high recurrence and resistance to current combination therapies including oncotomy, radiotherapy and chemotherapy. Light has been shed in the recent years on the anticancer properties of cannabinoids from Cannabis sativa.
OBJECTIVE
: To determine whether cannabinoids alone or in combination with radiotherapy and/or chemotherapy inhibit tumour progression, induce cancer cell death, inhibit metastasis and invasiveness and the mechanisms that underlie these actions.
METHOD
: PubMed and Web of Science were used for a systemic search to find studies on the anticancer effects of natural cannabinoids on glioma cancer cells in vitro and/or in vivo.
RESULTS
: A total of 302 papers were identified, of which 14 studies were found to fit the inclusion criteria. 5 studies were conducted in vitro, 2 in vivo and 7 were both in vivo and in vitro. 3 studies examined the efficacy of CBD, THC and TMZ, 1 study examined CBD and radiation, 2 studies examined efficacy of THC only and 3 studies examined the efficacy of CBD only. 1 study examined the efficacy of CBD, THC and radiotherapy, 2 studies examined the combination of CBD and THC and 2 more studies examined the efficacy of CBD and TMZ.
CONCLUSION
: The evidence in this systematic review leads to the conclusion that cannabinoids possess anticancer potencies against glioma cells, however this effect varies with the combinations and dosages used. Studies so far were conducted on cells in culture and on mice as well as a small number of studies that were conducted on humans. Hence in order to have more accurate results, higher quality studies mainly including human clinical trials with larger sample sizes are necessitated urgently for GBM treatment.”

 “Cannabidiol (CBD) has been shown to slow cancer cell growth and is toxic to human glioblastoma cell lines. Thus, CBD could be an effective therapeutic for glioblastoma.
“Cannabidiol (CBD) has been shown to slow cancer cell growth and is toxic to human glioblastoma cell lines. Thus, CBD could be an effective therapeutic for glioblastoma.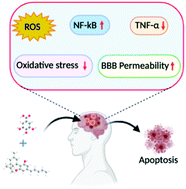
 “Preclinical data suggest some cannabinoids may exert antitumour effects against glioblastoma (GBM). Safety and preliminary efficacy of nabiximols oromucosal cannabinoid spray plus dose-intense temozolomide (DIT) was evaluated in patients with first recurrence of GBM.
“Preclinical data suggest some cannabinoids may exert antitumour effects against glioblastoma (GBM). Safety and preliminary efficacy of nabiximols oromucosal cannabinoid spray plus dose-intense temozolomide (DIT) was evaluated in patients with first recurrence of GBM. “Glioblastomas (GBMs) are aggressive brain tumors with frequent genetic alterations in TP53 and PTEN tumor suppressor genes rendering resistance to standard chemotherapeutics. Cannabinoid type 1 and 2 (CB1/CB2) receptor expression in GBMs and antitumor activity of cannabinoids in glioma cells and animal models, raised promises for a targeted treatment of these tumors. The susceptibility of human glioma cells to CB2-agonists and their mechanism of action are not fully elucidated. We determined CB1 and CB2 expression in 14 low-grade and 21 high-grade tumor biopsies, GBM-derived primary cultures and established cell lines. The non-selective CB receptor agonist WIN55,212-2 (but not its inactive enantiomer) or the CB2-selective agonist JWH133 induced apoptosis in patient-derived glioma cultures and five established glioma cell lines despite p53 and/or PTEN deficiency. Growth inhibitory efficacy of cannabinoids correlated with CB1/CB2 expression (EC50 WIN55,212-2: 7.36-15.70 µM, JWH133: 12.15-143.20 µM). Treatment with WIN55,212-2 or JWH133 led to activation of the apoptotic mitochondrial pathway and DNA fragmentation. Synthetic cannabinoid action was associated with the induction of autophagy and knockdown of autophagy genes augmented cannabinoid-induced apoptotic cell death. The high susceptibility of human glioblastoma cells to synthetic cannabinoids, despite genetic defects contributing to apoptosis resistance, makes cannabinoids promising anti-glioma therapeutics.”
“Glioblastomas (GBMs) are aggressive brain tumors with frequent genetic alterations in TP53 and PTEN tumor suppressor genes rendering resistance to standard chemotherapeutics. Cannabinoid type 1 and 2 (CB1/CB2) receptor expression in GBMs and antitumor activity of cannabinoids in glioma cells and animal models, raised promises for a targeted treatment of these tumors. The susceptibility of human glioma cells to CB2-agonists and their mechanism of action are not fully elucidated. We determined CB1 and CB2 expression in 14 low-grade and 21 high-grade tumor biopsies, GBM-derived primary cultures and established cell lines. The non-selective CB receptor agonist WIN55,212-2 (but not its inactive enantiomer) or the CB2-selective agonist JWH133 induced apoptosis in patient-derived glioma cultures and five established glioma cell lines despite p53 and/or PTEN deficiency. Growth inhibitory efficacy of cannabinoids correlated with CB1/CB2 expression (EC50 WIN55,212-2: 7.36-15.70 µM, JWH133: 12.15-143.20 µM). Treatment with WIN55,212-2 or JWH133 led to activation of the apoptotic mitochondrial pathway and DNA fragmentation. Synthetic cannabinoid action was associated with the induction of autophagy and knockdown of autophagy genes augmented cannabinoid-induced apoptotic cell death. The high susceptibility of human glioblastoma cells to synthetic cannabinoids, despite genetic defects contributing to apoptosis resistance, makes cannabinoids promising anti-glioma therapeutics.”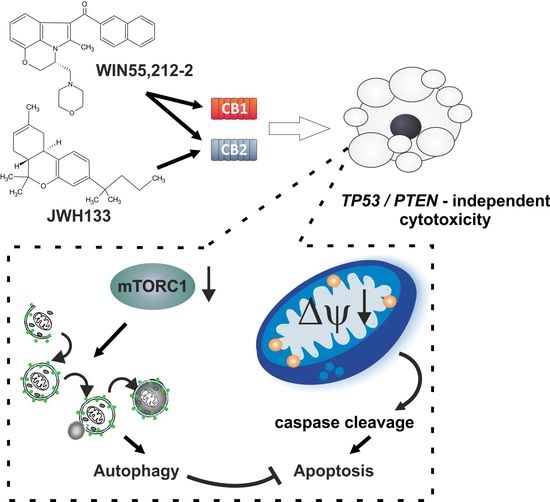
 “Glioblastoma is the most aggressive cancer among primary brain tumours. As with other cancers, the incidence of glioblastoma is increasing; despite modern therapies, the overall mean survival of patients post-diagnosis averages around 16 months, a figure that has not changed in many years. Cannabigerol (CBG) has only recently been reported to prevent the progression of certain carcinomas and has not yet been studied in glioblastoma. Here, we have compared the cytotoxic, apoptotic, and anti-invasive effects of the purified natural cannabinoid CBG together with CBD and THC on established differentiated glioblastoma tumour cells and glioblastoma stem cells. CBG and THC reduced the viability of both types of cells to a similar extent, whereas combining CBD with CBG was more efficient than with THC. CBD and CBG, both alone and in combination, induced caspase-dependent cell apoptosis, and there was no additive THC effect. Of note, CBG inhibited glioblastoma invasion in a similar manner to CBD and the chemotherapeutic temozolomide. We have demonstrated that THC has little added value in combined-cannabinoid glioblastoma treatment, suggesting that this psychotropic cannabinoid should be replaced with CBG in future clinical studies of glioblastoma therapy.”
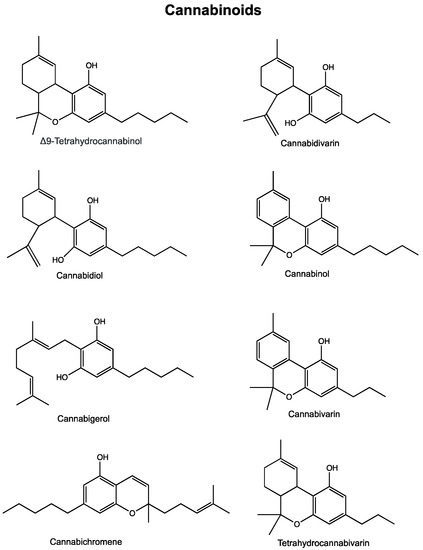
“Glioblastoma is the most aggressive cancer among primary brain tumours. As with other cancers, the incidence of glioblastoma is increasing; despite modern therapies, the overall mean survival of patients post-diagnosis averages around 16 months, a figure that has not changed in many years. Cannabigerol (CBG) has only recently been reported to prevent the progression of certain carcinomas and has not yet been studied in glioblastoma. Here, we have compared the cytotoxic, apoptotic, and anti-invasive effects of the purified natural cannabinoid CBG together with CBD and THC on established differentiated glioblastoma tumour cells and glioblastoma stem cells. CBG and THC reduced the viability of both types of cells to a similar extent, whereas combining CBD with CBG was more efficient than with THC. CBD and CBG, both alone and in combination, induced caspase-dependent cell apoptosis, and there was no additive THC effect. Of note, CBG inhibited glioblastoma invasion in a similar manner to CBD and the chemotherapeutic temozolomide. We have demonstrated that THC has little added value in combined-cannabinoid glioblastoma treatment, suggesting that this psychotropic cannabinoid should be replaced with CBG in future clinical studies of glioblastoma therapy.” “Cannabis has long been used for healing and recreation in several regions of the world. Over 400 bioactive constituents, including more than 100 phytocannabinoids, have been isolated from this plant. The non-psychoactive cannabidiol (CBD) and the psychoactive Δ9-tetrahydrocannabinol (Δ9-THC) are the major and widely studied constituents from this plant.
“Cannabis has long been used for healing and recreation in several regions of the world. Over 400 bioactive constituents, including more than 100 phytocannabinoids, have been isolated from this plant. The non-psychoactive cannabidiol (CBD) and the psychoactive Δ9-tetrahydrocannabinol (Δ9-THC) are the major and widely studied constituents from this plant. “Providers need to be better equipped to discuss medical cannabis with patients even if they are not willing to prescribe it. The oncology community would be well served to ensure that providers are aware of existing cannabis research and are able to incorporate it into their communications with patients instead of leaving patients to figure out medical cannabis on their own.”
“Providers need to be better equipped to discuss medical cannabis with patients even if they are not willing to prescribe it. The oncology community would be well served to ensure that providers are aware of existing cannabis research and are able to incorporate it into their communications with patients instead of leaving patients to figure out medical cannabis on their own.”