“Cannabis sativa L. is an annual herbaceous dioecious plant which was first cultivated by agricultural human societies in Asia. Over the period of time, various parts of the plant like leaf, flower, and seed were used for recreational as well as therapeutic purposes. The main chemical components of Cannabis sativa are termed as cannabinoids, among them the key psychoactive constituent is Δ-9-tetrahydrocannabinol and cannabidiol (CBD) as active nonpsychotic constituent. Upon doing extensive literature review, it was found that cannabis has been widely studied for a number of disorders. Very recently, a pure CBD formulation, named Epidiolex, got a green flag from both United States Food and Drug Administration and Drug Enforcement Administration for 2 rare types of epilepsies. This laid a milestone in medical cannabis research.
This review intends to give a basic and extensive assessment, from past till present, of the ethnological, plant, chemical, pharmacological, and legal aspects of C. sativa. Further, this review contemplates the evidence the studies obtained of cannabis components on Alzheimer’s, Parkinson’s, amyotrophic lateral sclerosis, multiple sclerosis, emesis, epilepsy, chronic pain, and cancer as a cytotoxic agent as well as a palliative therapy. The assessment in this study was done by reviewing in extensive details from studies on historical importance, ethnopharmacological aspects, and legal grounds of C. sativa from extensive literature available on the scientific databases, with a vision for elevating further pharmaceutical research to investigate its total potential as a therapeutic agent.”
https://pubmed.ncbi.nlm.nih.gov/34676349/
“This study has analyzed and reviewed the historical, botanical, chemical, ethnopharmacological, and legal aspects of C. sativa from the first human use to the present medical applications with an analysis of its multiple therapeutic applications for various diseased conditions in the contemporary scientific context. There is an abundance of support for its several medicative uses as well as a possible benefit in various diseased conditions. Extensive pharmacological examination is still needed to better understand the clinical significance and uses of active cannabinoids in the treatment and prevention of chronic diseases. Also, cannabis can be chemically standardized and under prescription can be used. With the majority of the United States currently legalizing medicinal cannabis and/or restricted CBD-only use, physicians need to be educated on the history and correct clinical use of cannabis, as a result of which patients can know more and more about possible treatment utilizing cannabis. Medical cannabis has shown to have clinical efficacy in our past, and in present, data show its therapeutic effects. Extensive research in the field of cannabis can be very fruitful for the medicine world.”

 “Background: Children with cancer are increasingly using cannabis therapeutically.
“Background: Children with cancer are increasingly using cannabis therapeutically. “Pain prevalence among adults in the United States has increased 25% over the past two decades, resulting in high health-care costs and impacts to patient quality of life. In the last 30 years, our understanding of pain circuits and (intra)cellular mechanisms has grown exponentially, but this understanding has not yet resulted in improved therapies. Options for pain management are limited. Many analgesics have poor efficacy and are accompanied by severe side effects such as addiction, resulting in a devastating opioid abuse and overdose epidemic. These problems have encouraged scientists to identify novel molecular targets and develop alternative pain therapeutics.
“Pain prevalence among adults in the United States has increased 25% over the past two decades, resulting in high health-care costs and impacts to patient quality of life. In the last 30 years, our understanding of pain circuits and (intra)cellular mechanisms has grown exponentially, but this understanding has not yet resulted in improved therapies. Options for pain management are limited. Many analgesics have poor efficacy and are accompanied by severe side effects such as addiction, resulting in a devastating opioid abuse and overdose epidemic. These problems have encouraged scientists to identify novel molecular targets and develop alternative pain therapeutics. “HIV-associated neurocognitive disorders (HAND) persist despite the advent of antiretroviral therapy (ART), suggesting underlying systemic and central nervous system (CNS) inflammatory mechanisms.
“HIV-associated neurocognitive disorders (HAND) persist despite the advent of antiretroviral therapy (ART), suggesting underlying systemic and central nervous system (CNS) inflammatory mechanisms. “Purpose:
“Purpose: 
 “Interest in CBG (cannabigerol) has been growing in the past few years, due to its anti-inflammatory properties and other therapeutic benefits.
“Interest in CBG (cannabigerol) has been growing in the past few years, due to its anti-inflammatory properties and other therapeutic benefits. 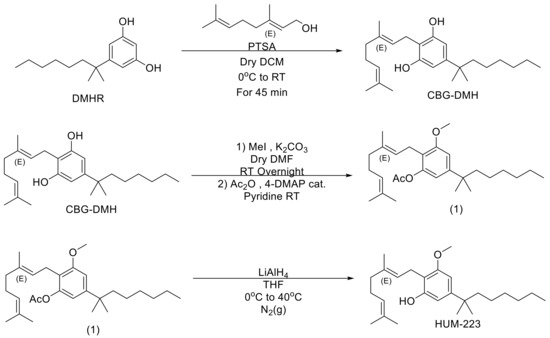
 “Acute respiratory distress syndrome (ARDS) is a life-threatening clinical syndrome whose potential to become one of the most grievous challenges of the healthcare system evidenced by the COVID-19 pandemic. Considering the lack of target-specific treatment for ARDS, it is absolutely exigent to have an effective therapeutic modality to reduce hospitalization and mortality rate as well as to improve quality of life and outcomes for ARDS patients. ARDS is a systemic inflammatory disease starting with the pulmonary system and involves all other organs in a morbid bidirectional fashion. Mounting evidence including our findings supporting the notion that cannabinoids have potential to be targeted as regulatory therapeutic modalities in the treatment of inflammatory diseases. Therefore, it is plausible to test their capabilities as alternative therapies in the treatment of ARDS. In this study, we investigated the potential protective effects of cannabichromene (CBC) in an experimental model of ARDS.
“Acute respiratory distress syndrome (ARDS) is a life-threatening clinical syndrome whose potential to become one of the most grievous challenges of the healthcare system evidenced by the COVID-19 pandemic. Considering the lack of target-specific treatment for ARDS, it is absolutely exigent to have an effective therapeutic modality to reduce hospitalization and mortality rate as well as to improve quality of life and outcomes for ARDS patients. ARDS is a systemic inflammatory disease starting with the pulmonary system and involves all other organs in a morbid bidirectional fashion. Mounting evidence including our findings supporting the notion that cannabinoids have potential to be targeted as regulatory therapeutic modalities in the treatment of inflammatory diseases. Therefore, it is plausible to test their capabilities as alternative therapies in the treatment of ARDS. In this study, we investigated the potential protective effects of cannabichromene (CBC) in an experimental model of ARDS. “The global incidence of respiratory diseases and complications is increasing. Therefore, new methods of treatment, as well as prevention, need to be investigated.
“The global incidence of respiratory diseases and complications is increasing. Therefore, new methods of treatment, as well as prevention, need to be investigated.  “Aims: Characterizing cannabinoid receptors (CBRs) expressed in Ewing sarcoma (EWS) cell lines as potential targets for anti-cancer drug development.
“Aims: Characterizing cannabinoid receptors (CBRs) expressed in Ewing sarcoma (EWS) cell lines as potential targets for anti-cancer drug development. “Cancer, as a mysterious and complex disease, has a multi-stage molecular process that uses the cellular molecular machine and multiple signaling pathways to its advantage. Cannabinoids, as terpenophenolic compounds and their derivatives, showed influences on immune system responses, inflammation, and cell growth that have sparked a growing interest in exploring their effects on cancer cell fate, as well. A large body of evidence in experimental models indicating the involvement of cannabinoids and their related receptors in cancer cell growth, development, and fate. In accordance, the present study provided insights regarding the strengths and limits of cannabinoids and their receptors in critical steps of tumorigenesis and its underlying molecular pathways such as; cancer cell proliferation, type of cell death pathway, angiogenesis, invasion, metastasis and, immune system response. Based on the results of the present study and due to the contribution of cannabinoids in various cancer cell growth control processes, these compounds cancer can be considered worthwhile in finding new alternatives for cancer therapy.”
“Cancer, as a mysterious and complex disease, has a multi-stage molecular process that uses the cellular molecular machine and multiple signaling pathways to its advantage. Cannabinoids, as terpenophenolic compounds and their derivatives, showed influences on immune system responses, inflammation, and cell growth that have sparked a growing interest in exploring their effects on cancer cell fate, as well. A large body of evidence in experimental models indicating the involvement of cannabinoids and their related receptors in cancer cell growth, development, and fate. In accordance, the present study provided insights regarding the strengths and limits of cannabinoids and their receptors in critical steps of tumorigenesis and its underlying molecular pathways such as; cancer cell proliferation, type of cell death pathway, angiogenesis, invasion, metastasis and, immune system response. Based on the results of the present study and due to the contribution of cannabinoids in various cancer cell growth control processes, these compounds cancer can be considered worthwhile in finding new alternatives for cancer therapy.”