 “Decline in cognitive performance, an aspect of the normal aging process, is influenced by the endocannabinoid system (ECS). Cannabinoid receptor 1 (CB1) signaling diminishes with advancing age in specific brain regions that regulate learning and memory and abolishing CB1 receptor signaling accelerates cognitive aging in mice.
“Decline in cognitive performance, an aspect of the normal aging process, is influenced by the endocannabinoid system (ECS). Cannabinoid receptor 1 (CB1) signaling diminishes with advancing age in specific brain regions that regulate learning and memory and abolishing CB1 receptor signaling accelerates cognitive aging in mice.
We recently demonstrated that prolonged exposure to low dose (3 mg/kg/day) Δ9-tetrahydrocannabinol (THC) improved the cognitive performances in old mice on par with young untreated mice. Here we investigated the potential influence of cannabidiol (CBD) on this THC effect, because preclinical and clinical studies indicate that the combination of THC and CBD often exhibits an enhanced therapeutic effect compared to THC alone.
We first tested the effectiveness of a lower dose (1 mg/kg/day) THC, and then the efficacy of the combination of THC and CBD in 1:1 ratio, same as in the clinically approved medicine Sativex®. Our findings reveal that a 1 mg/kg/day THC dose still effectively improved spatial learning in aged mice. However, a 1:1 combination of THC and CBD failed to do so.
The presence of CBD induced temporal changes in THC metabolism ensuing in a transient elevation of blood THC levels. However, as CBD metabolizes, the inhibitory effect on THC metabolism was alleviated, causing a rapid clearance of THC. Thus, the beneficial effects of THC seemed to wane off more swiftly in the presence of CBD, due to these metabolic effects.
The findings indicate that THC-treatment alone is more efficient to improve spatial learning in aged mice than the 1:1 combination of THC and CBD.”
https://pubmed.ncbi.nlm.nih.gov/34526890/
“In conclusion, our observations indicate that 1 mg/kg/day THC dose is still effective in improving the spatial learning in aged mice. With regard to the efficacy, THC-alone has proved to be more efficient in improving spatial learning in aged mice than its 1:1 combination with CBD. However, the possibility of THC/CBD being efficient in other ratios or at the earliest time-points, like immediately after the treatment cease, cannot be negated. Possibly, reducing the dose of CBD may improve the efficacy of the THC/CBD combination.”
https://www.frontiersin.org/articles/10.3389/fnagi.2021.718850/full
 “Interest in CBG (cannabigerol) has been growing in the past few years, due to its anti-inflammatory properties and other therapeutic benefits.
“Interest in CBG (cannabigerol) has been growing in the past few years, due to its anti-inflammatory properties and other therapeutic benefits. 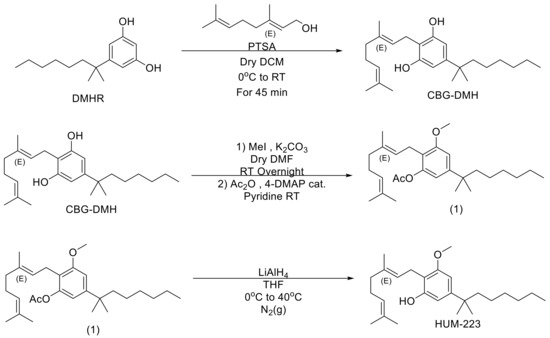

 “Beta-Caryophyllene (BCP), a naturally occurring sesquiterpene abundantly found in cloves, hops, and cannabis, is the active candidate of a relatively new group of vascular-inhibiting compounds that aim to block existing tumor blood vessels.
“Beta-Caryophyllene (BCP), a naturally occurring sesquiterpene abundantly found in cloves, hops, and cannabis, is the active candidate of a relatively new group of vascular-inhibiting compounds that aim to block existing tumor blood vessels.  “Acute respiratory distress syndrome (ARDS) is a life-threatening clinical syndrome whose potential to become one of the most grievous challenges of the healthcare system evidenced by the COVID-19 pandemic. Considering the lack of target-specific treatment for ARDS, it is absolutely exigent to have an effective therapeutic modality to reduce hospitalization and mortality rate as well as to improve quality of life and outcomes for ARDS patients. ARDS is a systemic inflammatory disease starting with the pulmonary system and involves all other organs in a morbid bidirectional fashion. Mounting evidence including our findings supporting the notion that cannabinoids have potential to be targeted as regulatory therapeutic modalities in the treatment of inflammatory diseases. Therefore, it is plausible to test their capabilities as alternative therapies in the treatment of ARDS. In this study, we investigated the potential protective effects of cannabichromene (CBC) in an experimental model of ARDS.
“Acute respiratory distress syndrome (ARDS) is a life-threatening clinical syndrome whose potential to become one of the most grievous challenges of the healthcare system evidenced by the COVID-19 pandemic. Considering the lack of target-specific treatment for ARDS, it is absolutely exigent to have an effective therapeutic modality to reduce hospitalization and mortality rate as well as to improve quality of life and outcomes for ARDS patients. ARDS is a systemic inflammatory disease starting with the pulmonary system and involves all other organs in a morbid bidirectional fashion. Mounting evidence including our findings supporting the notion that cannabinoids have potential to be targeted as regulatory therapeutic modalities in the treatment of inflammatory diseases. Therefore, it is plausible to test their capabilities as alternative therapies in the treatment of ARDS. In this study, we investigated the potential protective effects of cannabichromene (CBC) in an experimental model of ARDS. “The global incidence of respiratory diseases and complications is increasing. Therefore, new methods of treatment, as well as prevention, need to be investigated.
“The global incidence of respiratory diseases and complications is increasing. Therefore, new methods of treatment, as well as prevention, need to be investigated.  “Given the abundancy of angiotensin converting enzyme 2 (ACE-2) receptors density, beyond the lung, the intestine is considered as an alternative site of infection and replication for severe acute respiratory syndrome by coronavirus type 2 (SARS-CoV-2).
“Given the abundancy of angiotensin converting enzyme 2 (ACE-2) receptors density, beyond the lung, the intestine is considered as an alternative site of infection and replication for severe acute respiratory syndrome by coronavirus type 2 (SARS-CoV-2).  “This study consists of a brief psychological intervention, which uses Self-Regulation Therapy (SRT, procedure based on suggestion and classical conditioning), to improve coping with stress and emotionality by reproducing the positive effects of illegal drugs: cannabis, cocaine, ecstasy.
“This study consists of a brief psychological intervention, which uses Self-Regulation Therapy (SRT, procedure based on suggestion and classical conditioning), to improve coping with stress and emotionality by reproducing the positive effects of illegal drugs: cannabis, cocaine, ecstasy.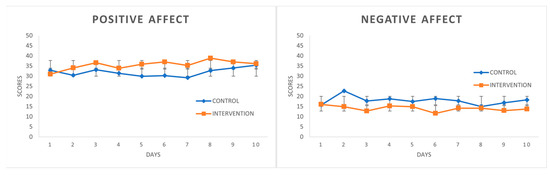
 “Conventional lung cancer treatments include surgery, chemotherapy and radiotherapy; however, these treatments are often poorly tolerated by patients. Cannabinoids have been studied for use as a primary cancer treatment. Cannabinoids, which are chemically similar to our own body’s endocannabinoids, can interact with signalling pathways to control the fate of cells, including cancer cells. We present a patient who declined conventional lung cancer treatment. Without the knowledge of her clinicians, she chose to self-administer ‘cannabidiol (CBD) oil’ orally 2-3 times daily. Serial imaging shows that her cancer reduced in size progressively from 41 mm to 10 mm over a period of 2.5 years. Previous studies have failed to agree on the usefulness of cannabinoids as a cancer treatment. This case appears to demonstrate a possible benefit of ‘CBD oil’ intake that may have resulted in the observed tumour regression. The use of cannabinoids as a potential cancer treatment justifies further research.”
“Conventional lung cancer treatments include surgery, chemotherapy and radiotherapy; however, these treatments are often poorly tolerated by patients. Cannabinoids have been studied for use as a primary cancer treatment. Cannabinoids, which are chemically similar to our own body’s endocannabinoids, can interact with signalling pathways to control the fate of cells, including cancer cells. We present a patient who declined conventional lung cancer treatment. Without the knowledge of her clinicians, she chose to self-administer ‘cannabidiol (CBD) oil’ orally 2-3 times daily. Serial imaging shows that her cancer reduced in size progressively from 41 mm to 10 mm over a period of 2.5 years. Previous studies have failed to agree on the usefulness of cannabinoids as a cancer treatment. This case appears to demonstrate a possible benefit of ‘CBD oil’ intake that may have resulted in the observed tumour regression. The use of cannabinoids as a potential cancer treatment justifies further research.” “Cancer, as a mysterious and complex disease, has a multi-stage molecular process that uses the cellular molecular machine and multiple signaling pathways to its advantage. Cannabinoids, as terpenophenolic compounds and their derivatives, showed influences on immune system responses, inflammation, and cell growth that have sparked a growing interest in exploring their effects on cancer cell fate, as well. A large body of evidence in experimental models indicating the involvement of cannabinoids and their related receptors in cancer cell growth, development, and fate. In accordance, the present study provided insights regarding the strengths and limits of cannabinoids and their receptors in critical steps of tumorigenesis and its underlying molecular pathways such as; cancer cell proliferation, type of cell death pathway, angiogenesis, invasion, metastasis and, immune system response. Based on the results of the present study and due to the contribution of cannabinoids in various cancer cell growth control processes, these compounds cancer can be considered worthwhile in finding new alternatives for cancer therapy.”
“Cancer, as a mysterious and complex disease, has a multi-stage molecular process that uses the cellular molecular machine and multiple signaling pathways to its advantage. Cannabinoids, as terpenophenolic compounds and their derivatives, showed influences on immune system responses, inflammation, and cell growth that have sparked a growing interest in exploring their effects on cancer cell fate, as well. A large body of evidence in experimental models indicating the involvement of cannabinoids and their related receptors in cancer cell growth, development, and fate. In accordance, the present study provided insights regarding the strengths and limits of cannabinoids and their receptors in critical steps of tumorigenesis and its underlying molecular pathways such as; cancer cell proliferation, type of cell death pathway, angiogenesis, invasion, metastasis and, immune system response. Based on the results of the present study and due to the contribution of cannabinoids in various cancer cell growth control processes, these compounds cancer can be considered worthwhile in finding new alternatives for cancer therapy.” “Anxiety and depressive disorders are highly prevalent. Patients are increasingly using medicinal cannabis products to treat these disorders, but little is known about the effects of medicinal cannabis use on symptoms of anxiety and depression.
“Anxiety and depressive disorders are highly prevalent. Patients are increasingly using medicinal cannabis products to treat these disorders, but little is known about the effects of medicinal cannabis use on symptoms of anxiety and depression.  “Decline in cognitive performance, an aspect of the normal aging process, is influenced by the endocannabinoid system (ECS). Cannabinoid receptor 1 (CB1) signaling diminishes with advancing age in specific brain regions that regulate learning and memory and abolishing CB1 receptor signaling accelerates cognitive aging in mice.
“Decline in cognitive performance, an aspect of the normal aging process, is influenced by the endocannabinoid system (ECS). Cannabinoid receptor 1 (CB1) signaling diminishes with advancing age in specific brain regions that regulate learning and memory and abolishing CB1 receptor signaling accelerates cognitive aging in mice.