 “∆ 9 -Tetrahydrocannabinol (∆9 -THC), the active phytocannabinoid in cannabis, is virtually an adjunct to the endogenous endocannabinoid signaling system.
“∆ 9 -Tetrahydrocannabinol (∆9 -THC), the active phytocannabinoid in cannabis, is virtually an adjunct to the endogenous endocannabinoid signaling system.
By interacting with G-protein-coupled receptors CB1 and CB2, ∆9 -THC affects peripheral and central circulation by lowering sympathetic activity, altering gene expression, cell proliferation, and differentiation, decreasing leukocyte migration, modulating neurotransmitter release thereby modulating cardiovascular functioning, tumorigenesis, immune responses, behavioral and locomotory activities respectively.
∆ 9 -THC is effective in suppressing chemotherapy-induced vomiting, retards malignant tumor growth, inhibits metastasis, and promotes apoptosis. Other mechanisms involved are targeting cell cycle at the G2-M phase in human breast cancer, downregulation of E2F transcription factor 1 (E2F1) in human glioblastoma multiforme, and stimulation of ER stress-induced autophagy.
∆ 9 -THC also plays a role in ameliorating neuroinflammation, excitotoxicity, neuroplasticity, trauma, and stroke and is associated with reliving childhood epilepsy, brain trauma, and neurodegenerative diseases.
∆9 -THC via CB1 receptors affects nociception, emotion, memory, and reduces neuronal excitability and excitotoxicity in epilepsy. It also increases renal blood flow, reduces intraocular pressure via a sympathetic pathway, and modulates hormonal release, thereby decreasing the reproductive function and increasing glucose metabolism.
Versatile medical marijuana has stimulated abundant research demonstrating substantial therapeutic promise, suggesting the possibilities of first-in-class drugs in diverse therapeutic segments. In this review, we represent the current pharmacological status of the phytocannabinoid, ∆ 9 -THC, and synthetic analogs in cancer, cardiovascular, and neurodegenerative disorders.”
https://pubmed.ncbi.nlm.nih.gov/33001012/
https://www.eurekaselect.com/186455/article


 “∆ 9 -Tetrahydrocannabinol (∆9 -THC), the active phytocannabinoid in cannabis, is virtually an adjunct to the endogenous endocannabinoid signaling system.
“∆ 9 -Tetrahydrocannabinol (∆9 -THC), the active phytocannabinoid in cannabis, is virtually an adjunct to the endogenous endocannabinoid signaling system. “Pancreatic cancer (PC) is related to lifestyle risks, chronic inflammation, and germline mutations in BRCA1/2, ATM, MLH1, TP53, or CDKN2A. Surgical resection and adjuvant chemotherapy are the main therapeutic strategies but are less effective in patients with high-grade tumors.
“Pancreatic cancer (PC) is related to lifestyle risks, chronic inflammation, and germline mutations in BRCA1/2, ATM, MLH1, TP53, or CDKN2A. Surgical resection and adjuvant chemotherapy are the main therapeutic strategies but are less effective in patients with high-grade tumors.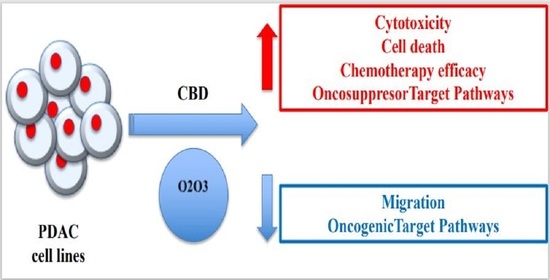
 “Prostate cancer is a major cause of death among men worldwide.
“Prostate cancer is a major cause of death among men worldwide. “Objective: Cannabinoids are able to reduce tumor growth in xenograft models, but their therapeutic potential as anti-cancer drugs in humans is unclear yet. In vitro studies of the effect of cannabinoids on cancer cells are often carried out in absence of serum or in low serum concentration (i.e. 0.5% serum), conditions that limit cellular growth and therefore can increase the response of cells to additional challenges such as the presence of cannabinoids. However, the tumor microenvironment can be teaming with growth factors. In this study we assessed the viability and proliferation of cancer cells treated with cannabidiol in presence of a serum concentration that commonly sustains cell growth (10% serum).
“Objective: Cannabinoids are able to reduce tumor growth in xenograft models, but their therapeutic potential as anti-cancer drugs in humans is unclear yet. In vitro studies of the effect of cannabinoids on cancer cells are often carried out in absence of serum or in low serum concentration (i.e. 0.5% serum), conditions that limit cellular growth and therefore can increase the response of cells to additional challenges such as the presence of cannabinoids. However, the tumor microenvironment can be teaming with growth factors. In this study we assessed the viability and proliferation of cancer cells treated with cannabidiol in presence of a serum concentration that commonly sustains cell growth (10% serum).
 “In this work, we evaluated, for the first time, the antitumor effect of cannabidiol (CBD) as monotherapy and in combination with conventional chemotherapeutics in ovarian cancer and developed PLGA-microparticles as CBD carriers to optimize its anticancer activity.
“In this work, we evaluated, for the first time, the antitumor effect of cannabidiol (CBD) as monotherapy and in combination with conventional chemotherapeutics in ovarian cancer and developed PLGA-microparticles as CBD carriers to optimize its anticancer activity.


 “Astrocytomas, the most prevalent primary brain tumors, can be divided by histology and malignancy levels into four following types: pilocytic astrocytoma (grade I), diffuse fibrillary astrocytoma (grade II), anaplastic astrocytoma (grade III), and glioblastoma multiforme (grade IV). For high grade astrocytomas (grade III and grade IV), blood vessels formation is considered as the most important property.
“Astrocytomas, the most prevalent primary brain tumors, can be divided by histology and malignancy levels into four following types: pilocytic astrocytoma (grade I), diffuse fibrillary astrocytoma (grade II), anaplastic astrocytoma (grade III), and glioblastoma multiforme (grade IV). For high grade astrocytomas (grade III and grade IV), blood vessels formation is considered as the most important property.