
“The Cannabis plant contains over 100 phytocannabinoids and hundreds of other components. The biological effects and interplay of these Cannabis compounds are not fully understood and yet influence the plant’s therapeutic effects.
Here we assessed the antitumor effects of whole Cannabis extracts, which contained significant amounts of differing phytocannabinoids, on different cancer lines from various tumor origins.
Our results show that specific Cannabis extracts impaired the survival and proliferation of cancer cell lines as well as induced apoptosis.
Our findings showed that pure (-)-Δ9–trans-tetrahydrocannabinol (Δ9-THC) did not produce the same effects on these cell lines as the whole Cannabis extracts. Furthermore, Cannabis extracts with similar amounts of Δ9-THC produced significantly different effects on the survival of specific cancer cells.
In addition, we demonstrated that specific Cannabis extracts may selectively and differentially affect cancer cells and differing cancer cell lines from the same organ origin. We also found that cannabimimetic receptors were differentially expressed among various cancer cell lines and suggest that this receptor diversity may contribute to the heterogeneous effects produced by the differing Cannabis extracts on each cell line.
Our overall findings indicate that the effect of a Cannabis extract on a specific cancer cell line relies on the extract’s composition as well as on certain characteristics of the targeted cells.”
http://www.oncotarget.com/index.php?journal=oncotarget&page=article&op=view&path[]=26983
“Many previous reports highlight and demonstrate the anti-tumor effects of cannabinoids. In the last decade, accumulating evidence has indicated that phytocannabinoids might have antitumor properties. A number of in vitro and in vivo studies have demonstrated the effects of phytocannabinoids on tumor progression by interrupting several characteristic features of cancer. These studies suggest that specific cannabinoids such as Δ9-THC and CBD induce apoptosis and inhibit proliferation in various cancer cell lines.”
http://www.oncotarget.com/index.php?journal=oncotarget&page=article&op=view&path%5B%5D=26983&path%5B%5D=85698
https://pubmed.ncbi.nlm.nih.gov/31289609/
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6609248/
 “Providers need to be better equipped to discuss medical cannabis with patients even if they are not willing to prescribe it. The oncology community would be well served to ensure that providers are aware of existing cannabis research and are able to incorporate it into their communications with patients instead of leaving patients to figure out medical cannabis on their own.”
“Providers need to be better equipped to discuss medical cannabis with patients even if they are not willing to prescribe it. The oncology community would be well served to ensure that providers are aware of existing cannabis research and are able to incorporate it into their communications with patients instead of leaving patients to figure out medical cannabis on their own.”
 “In recent years, and even more since its legalization in several jurisdictions, cannabis and the endocannabinoid system have received an increasing amount of interest related to their potential exploitation in clinical settings. Cannabinoids have been suggested and shown to be effective in the treatment of various conditions. In cancer, the endocannabinoid system is altered in numerous types of tumours and can relate to cancer prognosis and disease outcome. Additionally, cannabinoids display anticancer effects in several models by suppressing the proliferation, migration and/or invasion of cancer cells, as well as tumour angiogenesis. However, the therapeutic use of cannabinoids is currently limited to the treatment of symptoms and pain associated with chemotherapy, while their potential use as cytotoxic drugs in chemotherapy still requires validation in patients. Along with cannabinoids, cannabis contains several other compounds that have also been shown to exert anti-tumorigenic actions. The potential anti-cancer effects of cannabinoids, terpenes and flavonoids, present in cannabis, are explored in this literature review.”
“In recent years, and even more since its legalization in several jurisdictions, cannabis and the endocannabinoid system have received an increasing amount of interest related to their potential exploitation in clinical settings. Cannabinoids have been suggested and shown to be effective in the treatment of various conditions. In cancer, the endocannabinoid system is altered in numerous types of tumours and can relate to cancer prognosis and disease outcome. Additionally, cannabinoids display anticancer effects in several models by suppressing the proliferation, migration and/or invasion of cancer cells, as well as tumour angiogenesis. However, the therapeutic use of cannabinoids is currently limited to the treatment of symptoms and pain associated with chemotherapy, while their potential use as cytotoxic drugs in chemotherapy still requires validation in patients. Along with cannabinoids, cannabis contains several other compounds that have also been shown to exert anti-tumorigenic actions. The potential anti-cancer effects of cannabinoids, terpenes and flavonoids, present in cannabis, are explored in this literature review.”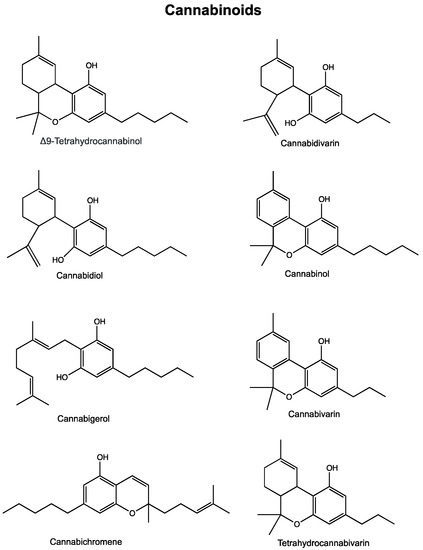
 “The recent announcement of marijuana legalization in Canada spiked many discussions about potential health benefits of Cannabis sativa.
“The recent announcement of marijuana legalization in Canada spiked many discussions about potential health benefits of Cannabis sativa.  “Patients with non-small cell lung cancer (NSCLC) develop resistance to antitumor agents by mechanisms that involve the epithelial-to-mesenchymal transition (EMT). This necessitates the development of new complementary drugs, e.g.,
“Patients with non-small cell lung cancer (NSCLC) develop resistance to antitumor agents by mechanisms that involve the epithelial-to-mesenchymal transition (EMT). This necessitates the development of new complementary drugs, e.g.,  “In recent years, the endocannabinoid system has received great interest as a potential therapeutic target in numerous pathological conditions.
“In recent years, the endocannabinoid system has received great interest as a potential therapeutic target in numerous pathological conditions. “Phytocannabinoids are unique terpenophenolic compounds predominantly produced in the glandular trichomes of the cannabis plant (Cannabis sativa L.). The delta-9- tetrahydrocannabinol (THC) is the main active constituent responsible for the plant’s psychoactive effect and, together with the non- psychoactive cannabidiol (CBD), the most investigated naturally occurring cannabinoid.
“Phytocannabinoids are unique terpenophenolic compounds predominantly produced in the glandular trichomes of the cannabis plant (Cannabis sativa L.). The delta-9- tetrahydrocannabinol (THC) is the main active constituent responsible for the plant’s psychoactive effect and, together with the non- psychoactive cannabidiol (CBD), the most investigated naturally occurring cannabinoid.

