 “”Medicinal cannabis” is defined as the use of cannabis-based products for the treatment of an illness. Investigations of cannabis compounds in psychiatric and neurological illnesses primarily focus on the major cannabinoids, cannabidiol (CBD) and Δ9-tetrahydrocannabinol (Δ9-THC), which are hypothesised to benefit multiple illnesses manifesting cognitive impairment, neurodegeneration and neuro-inflammation, as well as chronic pain, epilepsy and post-traumatic stress disorder, respectively.
“”Medicinal cannabis” is defined as the use of cannabis-based products for the treatment of an illness. Investigations of cannabis compounds in psychiatric and neurological illnesses primarily focus on the major cannabinoids, cannabidiol (CBD) and Δ9-tetrahydrocannabinol (Δ9-THC), which are hypothesised to benefit multiple illnesses manifesting cognitive impairment, neurodegeneration and neuro-inflammation, as well as chronic pain, epilepsy and post-traumatic stress disorder, respectively.
The cannabis plant contains >500 compounds, including terpenes responsible for the flavour and fragrance profiles of plants. Recently, research has begun providing evidence on the potential use of certain plant-derived terpenes in modern medicine, demonstrating anti-oxidant, anti-inflammatory, and neuroprotective effects of these compounds.
This review examined the effects of two key terpenes, pinene and linalool, on parameters relevant to neurological and psychiatric disorders, highlighting gaps in the literature and recommendations for future research into terpene therapeutics.
Overall, evidence is mostly limited to preclinical studies and well-designed clinical trials are lacking. Nevertheless, existing data suggests that pinene and linalool are relevant candidates for further investigation as novel medicines for illnesses, including stroke, ischemia, inflammatory and neuropathic pain (including migraine), cognitive impairment (relevant to Alzheimer’s disease and ageing), insomnia, anxiety, and depression.
Linalool and pinene influence multiple neurotransmitter, inflammatory and neurotrophic signals as well as behaviour, demonstrating psycho-activity (albeit non-intoxicating). Optimising the phytochemical profile of cannabis chemovars to yield therapeutic levels of beneficial terpenes and cannabinoids, such as linalool, pinene and CBD, could present a unique opportunity to discover novel medicines to treat psychiatric and neurological illnesses; however, further research is needed.”
https://pubmed.ncbi.nlm.nih.gov/34512404/
“Overall, it appears that the importance of the terpene profile of plants to humans extends further than mere olfactory and gustatory delight. Rather, these compounds have the potential for use as treatments for serious chronic neurological and psychiatric illnesses.”
https://www.frontiersin.org/articles/10.3389/fpsyt.2021.583211/full

 “Cannabinoids, active components of the plant
“Cannabinoids, active components of the plant  “The Endocannabinoid System (ECS) is primarily responsible for maintaining homeostasis, a balance in internal environment (temperature, mood, and immune system) and energy input and output in living, biological systems.
“The Endocannabinoid System (ECS) is primarily responsible for maintaining homeostasis, a balance in internal environment (temperature, mood, and immune system) and energy input and output in living, biological systems. 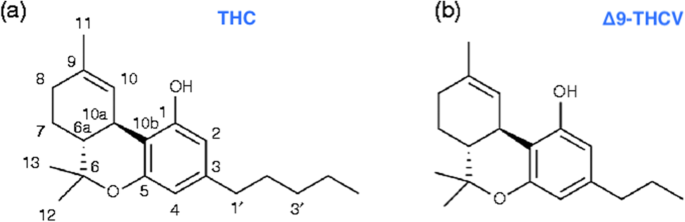 “Δ9-Tetrahydrocannabivarin (THCV) is a cannabis-derived compound with unique properties that set it apart from the more common cannabinoids, such as Δ9-tetrahydrocannabinol (THC). The main advantage of THCV over THC is the lack of psychoactive effects.
“Δ9-Tetrahydrocannabivarin (THCV) is a cannabis-derived compound with unique properties that set it apart from the more common cannabinoids, such as Δ9-tetrahydrocannabinol (THC). The main advantage of THCV over THC is the lack of psychoactive effects.  “Nowadays cardiovascular diseases (CVDs) are the major causes for the reduction of the quality of life.
“Nowadays cardiovascular diseases (CVDs) are the major causes for the reduction of the quality of life. “In humans, various sites like cannabinoid receptors (CBR) having a binding affinity with cannabinoids are distributed on the surface of different cell types, where endocannabinoids (ECs) and derivatives of fatty acid can bind. The binding of these substance(s) triggers the activation of specific receptors required for various physiological functions, including pain sensation, memory, and appetite.
“In humans, various sites like cannabinoid receptors (CBR) having a binding affinity with cannabinoids are distributed on the surface of different cell types, where endocannabinoids (ECs) and derivatives of fatty acid can bind. The binding of these substance(s) triggers the activation of specific receptors required for various physiological functions, including pain sensation, memory, and appetite. 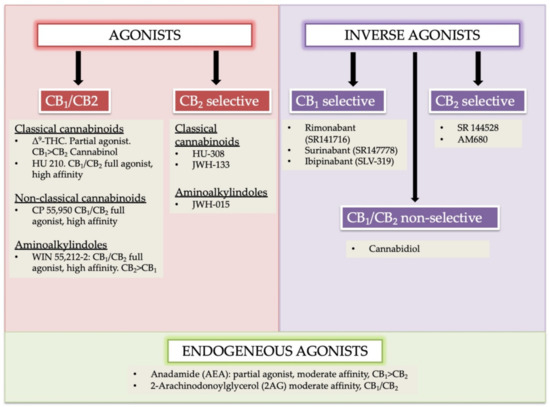
 “The cannabinoid, cannabidiol (CBD), is part of the plant’s natural defense system that when given to animals has many useful medicinal properties, including activity against cancer cells, modulation of the immune system, and efficacy in epilepsy.
“The cannabinoid, cannabidiol (CBD), is part of the plant’s natural defense system that when given to animals has many useful medicinal properties, including activity against cancer cells, modulation of the immune system, and efficacy in epilepsy.  “Venous Leg Ulcers are highly prevalent lower limb integumentary wounds that remain challenging to heal despite the use of evidence-based compression therapies. A multitude of adjuvant treatments have been studied but none have demonstrated enough efficacy to gain adoption into treatment guidelines.
“Venous Leg Ulcers are highly prevalent lower limb integumentary wounds that remain challenging to heal despite the use of evidence-based compression therapies. A multitude of adjuvant treatments have been studied but none have demonstrated enough efficacy to gain adoption into treatment guidelines.