 “Anticancer properties of non-psychoactive cannabinoid cannabidiol (CBD) have been demonstrated on tumors of different histogenesis. Different molecular targets for CBD were proposed, including cannabinoid receptors and some plasma membrane ion channels. Here we have shown that cell lines derived from acute lymphoblastic leukemia of T lineage (T-ALL), but not resting healthy T cells, are highly sensitive to CBD treatment. CBD effect does not depend on cannabinoid receptors or plasma membrane Ca2+-permeable channels. Instead, CBD directly targets mitochondria and alters their capacity to handle Ca2+. At lethal concentrations, CBD causes mitochondrial Ca2+ overload, stable mitochondrial transition pore formation and cell death. Our results suggest that CBD is an attractive candidate to be included into chemotherapeutic protocols for T-ALL treatment.”
“Anticancer properties of non-psychoactive cannabinoid cannabidiol (CBD) have been demonstrated on tumors of different histogenesis. Different molecular targets for CBD were proposed, including cannabinoid receptors and some plasma membrane ion channels. Here we have shown that cell lines derived from acute lymphoblastic leukemia of T lineage (T-ALL), but not resting healthy T cells, are highly sensitive to CBD treatment. CBD effect does not depend on cannabinoid receptors or plasma membrane Ca2+-permeable channels. Instead, CBD directly targets mitochondria and alters their capacity to handle Ca2+. At lethal concentrations, CBD causes mitochondrial Ca2+ overload, stable mitochondrial transition pore formation and cell death. Our results suggest that CBD is an attractive candidate to be included into chemotherapeutic protocols for T-ALL treatment.”
https://www.ncbi.nlm.nih.gov/pubmed/31611561
“Considering the pivotal role of mitochondria in oncogenic re-programming, CBD may be plausible candidate to be included into chemotherapeutic protocols.”

 “Cancer-related cachexia and anorexia syndrome (CACS) is a common phenomenon in cancer patients. Cannabis has been suggested to stimulate appetite but research on this issue has yielded mixed results. The current study aimed to evaluate the effect of dosage-controlled cannabis capsules on CACS in advanced cancer patients.
“Cancer-related cachexia and anorexia syndrome (CACS) is a common phenomenon in cancer patients. Cannabis has been suggested to stimulate appetite but research on this issue has yielded mixed results. The current study aimed to evaluate the effect of dosage-controlled cannabis capsules on CACS in advanced cancer patients.
 “Grade IV glioblastoma multiforme is a deadly disease, with a median survival of around 14 to 16 months. Maximal resection followed by adjuvant radiochemotherapy has been the mainstay of treatment since many years, although survival is only extended by a few months. In recent years, an increasing number of data from in vitro and in vivo research with cannabinoids, particularly with the non-intoxicating cannabidiol (CBD), point to their potential role as tumour-inhibiting agents. Herein, a total of nine consecutive patients with brain tumours are described as case series; all patients received CBD in a daily dose of 400 mg concomitantly to the standard therapeutic procedure of maximal resection followed by radiochemotherapy. By the time of the submission of this article, all but one patient are still alive with a mean survival time of 22.3 months (range=7-47 months). This is longer than what would have been expected.”
“Grade IV glioblastoma multiforme is a deadly disease, with a median survival of around 14 to 16 months. Maximal resection followed by adjuvant radiochemotherapy has been the mainstay of treatment since many years, although survival is only extended by a few months. In recent years, an increasing number of data from in vitro and in vivo research with cannabinoids, particularly with the non-intoxicating cannabidiol (CBD), point to their potential role as tumour-inhibiting agents. Herein, a total of nine consecutive patients with brain tumours are described as case series; all patients received CBD in a daily dose of 400 mg concomitantly to the standard therapeutic procedure of maximal resection followed by radiochemotherapy. By the time of the submission of this article, all but one patient are still alive with a mean survival time of 22.3 months (range=7-47 months). This is longer than what would have been expected.”
 “Little is known about the endocannabinoid (eCB) system in squamous cell carcinoma of the oral tongue (SCCOT). Here we have investigated, at the mRNA level, expression of genes coding for the components of the eCB system in tumour and non-malignant samples from SCCOT patients. Expression of NAPEPLD and PLA2G4E, coding for eCB anabolic enzymes, was higher in the tumour tissue than in non-malignant tissue. Among genes coding for eCB catabolic enzymes, expression of MGLL was lower in tumour tissue while PTGS2 was increased. It is concluded that the eCB system may be dysfunctional in SCCOT.”
“Little is known about the endocannabinoid (eCB) system in squamous cell carcinoma of the oral tongue (SCCOT). Here we have investigated, at the mRNA level, expression of genes coding for the components of the eCB system in tumour and non-malignant samples from SCCOT patients. Expression of NAPEPLD and PLA2G4E, coding for eCB anabolic enzymes, was higher in the tumour tissue than in non-malignant tissue. Among genes coding for eCB catabolic enzymes, expression of MGLL was lower in tumour tissue while PTGS2 was increased. It is concluded that the eCB system may be dysfunctional in SCCOT.” “The endogenous lipid metabolism network is associated with the occurrence and progression of malignancies.
“The endogenous lipid metabolism network is associated with the occurrence and progression of malignancies. “Indisputably, cancer is a global crisis that requires immediate intervention. Despite the use of conventional treatments over the past decades, it is acceptable to admit that these are expensive, invasive, associated with many side effects and, therefore, a reduced quality of life.
“Indisputably, cancer is a global crisis that requires immediate intervention. Despite the use of conventional treatments over the past decades, it is acceptable to admit that these are expensive, invasive, associated with many side effects and, therefore, a reduced quality of life.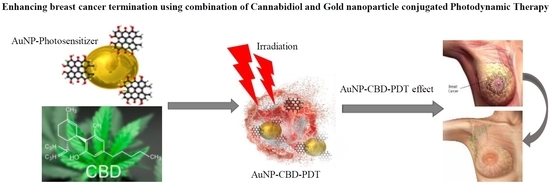
 “Cancer patients experience multiple symptoms throughout their illness, and some report benefit from the use of cannabis. There are concerns that many patients are accessing products inappropriate for their situation and potentially putting themselves at risk.
“Cancer patients experience multiple symptoms throughout their illness, and some report benefit from the use of cannabis. There are concerns that many patients are accessing products inappropriate for their situation and potentially putting themselves at risk. “Cannabinoid receptors have been detected in human gliomas and cannabinoids have been proposed as novel drug candidates in the treatment of brain tumors.
“Cannabinoid receptors have been detected in human gliomas and cannabinoids have been proposed as novel drug candidates in the treatment of brain tumors.